Introduction of CLED Agar
Table of Contents
CLED stands for cysteine, lactose, and electrolyte-deficient. CLED agar or medium is a differential culture medium used for the isolation and enumeration of bacteria in urine specimens from suspected cases of urinary tract infection (UTI). It favors the growth of all potential urinary pathogens as well as a number of contaminants such as diphtheroid, lactobacilli, and micrococci. Candida also enjoys this medium. This is a sole medium alternative for routine urine culture and inoculation of specimens in a combination of blood agar (BAP) and MacConkey agar (MAC) without compromising the quality and reducing the cost.

Composition of CLED Medium
The composition of CLED Medium is shown below in the table-
| S.No. | Ingredients | Amount |
| 1. | Peptone | 2 gm |
| 2. | “Lab Lemco” Powder (meat extract) | 1.5 gm |
| 3. | Tryptone | 2 gm |
| 4. | Lactose | 5 gm |
| 5. | L-Cysteine | 0.064 |
| 6. | Bromothymol blue | 0.01g |
| 7. | Agar | 7.5 gm |
| 8. | Distilled water | 500 ml |
| Final pH | 7.3 +/- 0.2 at 25°C |
Preparation of CLED Agar
CLED agar is available in dehydrated powder form by various manufacturers like Mast, Oxoid, Difco, Himedia, and so on.
- Follow the instructions of the manufacturer to prepare the medium, i.e., 18.06 dehydrated medium in 500mL distilled or deionized water, mix properly, and finally sterilize by autoclaving at 121°C for 15 minutes.
- After cooling to 50-55°C, mix well, and dispense it aseptically in sterile Petri dishes. Date the medium and give it a batch number.
- Store the plates at 2-8°C preferably in plastic bags to prevent loss of moisture.
Shelf life of CLED Medium
It can be used for several weeks, but should be free from any change in the appearance of the medium, showing contamination, deterioration, or alteration of pH.
Principle of CLED Agar
Reduced electrolyte prevents the swarming of Proteus, whereas Cystine promotes the formation of cystine-dependent dwarf colonies. Bromothymol blue is the indicator used in the agar; it changes to yellow in case of acid production during fermentation of lactose or changes to deep blue in case of alkalization. Lactose-fermenting bacteria build yellow colonies. Bacteria that decarboxylate L-Cystine cause an alkaline reaction and build deep blue colonies.
Colony Characteristics of CLED Agar
Typical Colony Morphology on CLED Agar is as follows:
| S.No. | Organisms | Colony Morphology |
| 1. | Staphylococcus aureus | Deep yellow colonies of uniform color |
| 2. | S. saprophyticus and other Coagulase Negative Staphylococci (CoNS) | Pale yellow to white colonies, more opaque than Enterococcus faecalis |
| 3. | Enterococci | Small yellow colonies, about 0.5mm in diameter |
| 4. | E. coli: Introduction, Identification Features, Keynotes, and Escherichia coli Footages Escherichia coli | Opaque yellow colonies with a slightly deeper yellow center |
| 5. | Klebsiella species | Large mucoid yellow to whitish-blue colonies |
| 6. | Proteus spp. | Translucent blue-grey colonies |
| 7. | Pseudomonas aeruginosa | Green colonies with typical matted surface and rough periphery |
Modifications of CLED Agar
- Note: CLED medium with modification i.e. CLED Medium with Andrade’s Indicator now uses most commonly.
- Composition is the same with a slight increment of Andrade’s indicator i.e. Andrade indicator (0.100 gm/L)
- CLED Medium is further modified by Bevis by incorporating Andrade’s indicator. This medium provides sharper differentiation between lactose-fermenters (LF) and lactose-non-fermenters (NLF). The addition of Andrade’s indicator enhances the appearance of the colony and aids in the identification of microorganisms. At different pH values, the color of the medium varies from the standard medium, which is well documented by Bevis.
Color of CLED Medium
The difference in color due to different pH effects is as follows-
| pH | Color |
| 7.4 | deep blue |
| 7.0 | bluish grey |
| 6.8 | pale grey |
| 6.6 | pinkish grey |
| 6.4 | bright red with a whitish tinge |
| 6.0 | bright red |
For better results, the medium should not be incubated for more than 24 hours because if lactose fermenters predominate, the entire medium may turn pink, masking the presence of non-lactose fermenters. Inoculate the medium immediately after urine collection.
Typical colony morphology on CLED Medium with Andrade’s Indicator
Typical colony morphology on CLED Medium with Andrade’s Indicator after incubation at 35-37°C for 18-24 hours is as follows:
| S.No | Microbes | Colony Characteristics |
| 1. | Klebsiella aerogenes ATCC 13048 | greyish green, mucoid |
| 2. | Escherichia coli ATCC 25922 | bright pink with a pink halo |
| 3. | Enterococcus faecalis ATCC 29212 | orange-yellow or greenish |
| 4. | Proteus mirabilis ATCC 25933 | blue-green |
| 5. | Staphylococcus aureus ATCC 25923 | golden-yellow |
| 6. | Streptococcus pyogenes ATCC 19615 | greyish green |
Proteus mirabilis green-blue colony on CLED medium
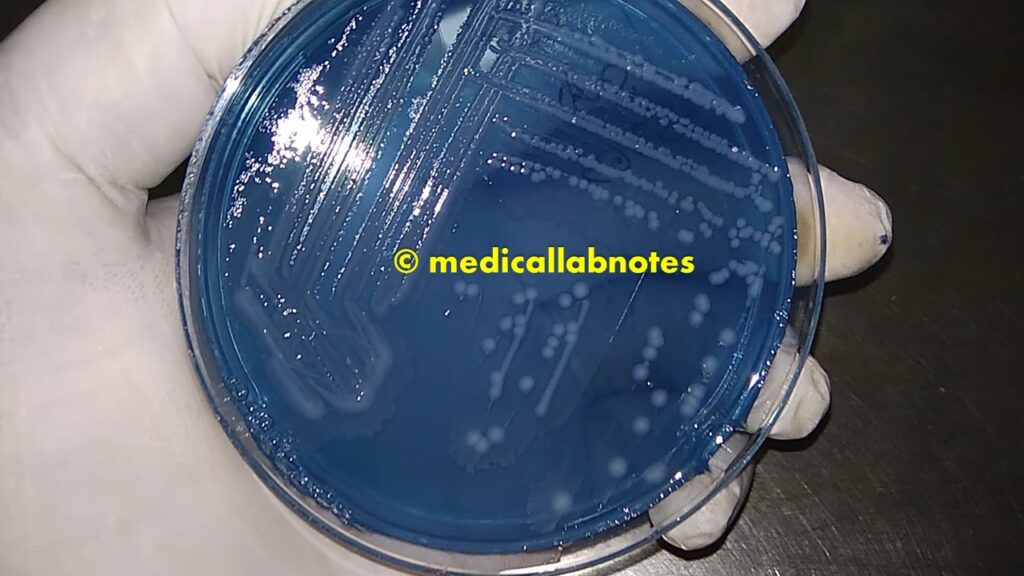
Staphylococcus aureus golden yellow colonies on CLED agar after 4 days of incubation

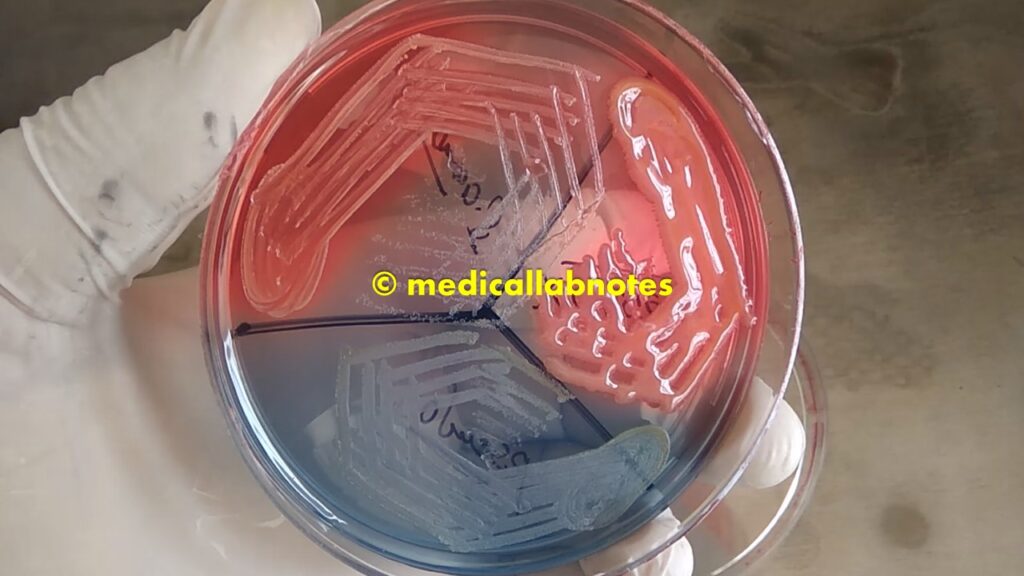
Mixed growth of microbes on CLED agar in urine culture, showing Candida and bacterial growth after 24 hours of incubation

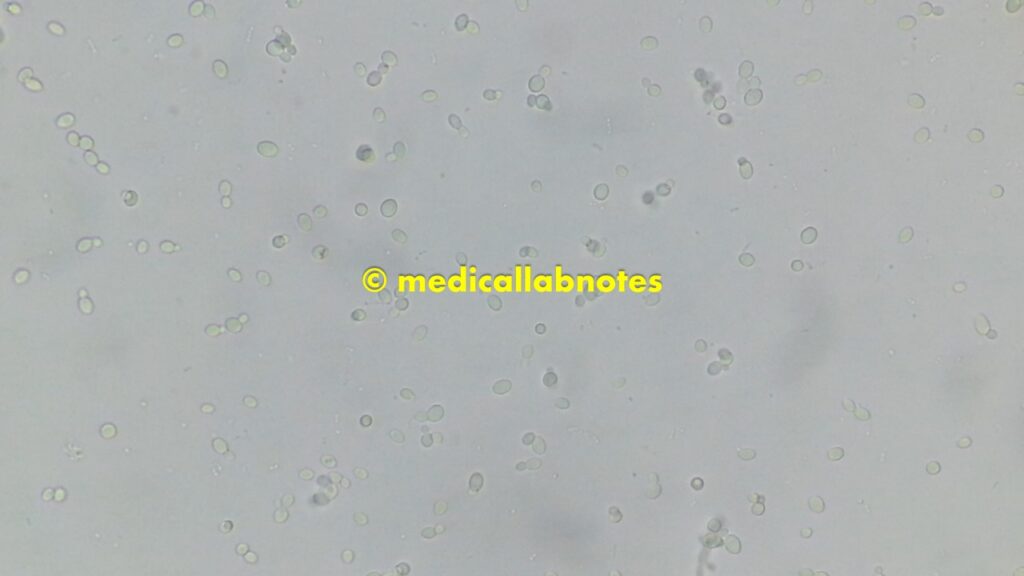
Saline microscopy of a mixed growth of microbes from the above culture plate, showing yeast cells and bacteria
Candida colony morphology on CLED agar

Gram-positive yeast cells of Candida

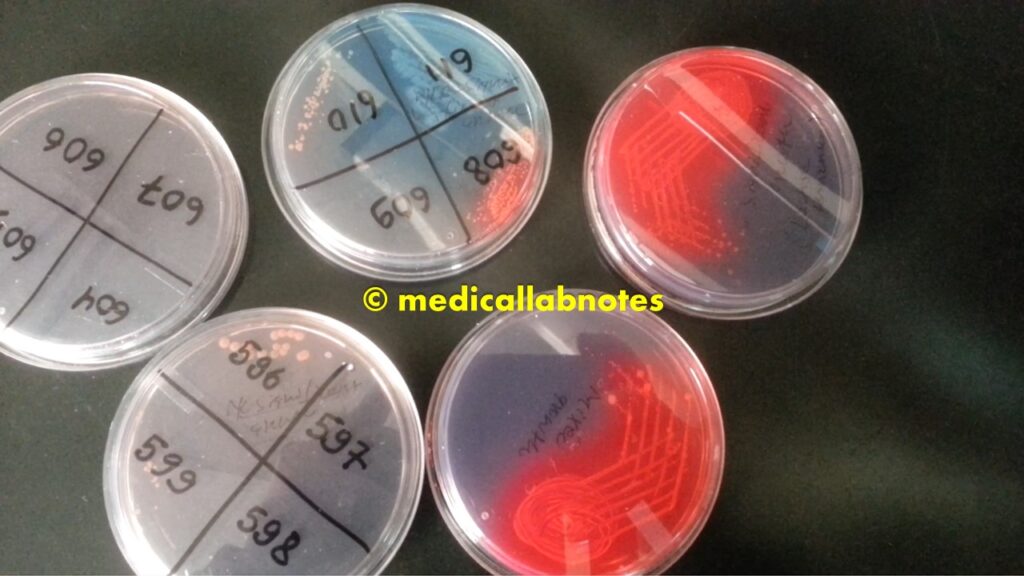
Various microbial growth on CLED agar plates of urine culture
Advantages of CLED Agar
(CLED Agar for Urine Culture)

- Good discrimination of gram-negative bacteria based on lactose fermentation and colony appearance.
- It inhibits the swarming of Proteus species (Proteus mirabilis and Proteus vulgaris are frequently involved in urinary tract infections) due to electrolyte deficiency.
- Relatively low cost as compared to the combined use of blood agar and MacConkey agar for urine culture.
Limitations of CLED Medium

- This medium is recommended for urine infections. Low urine count may be a result of antibiotic therapy and low pH of urine.
- Recovery depends on the urine count.
- Inoculate the medium immediately after urine collection.
- Shigella species may not grow on this medium.
- For better results, the medium should not be incubated for more than 24 hours because if lactose fermenters predominate, the entire medium may turn pink, masking the presence of non-lactose fermenters.
Keynotes on CLED Agar

- The color of the colony is directly affected by pH variance.
- The colony morphology of microbes may vary according to modifications of CLED media (e.g., use of Andrade’s indicator).
- It is one of the recommended culture media for urine culture, replacing the use of MacConkey and blood agar, which is also beneficial for the reduction of laboratory costs and labor.
- There is a need for trained staff to report CLED agar due to having growth of Gram-positive, negative, and even fungi, which is a common drawback of using CLED agar.
Further Readings
- Bailey & Scott’s Diagnostic Microbiology. Editors: Bettey A. Forbes, Daniel F. Sahm & Alice S. Weissfeld, 12th ed 2007, Publisher Elsevier.
- Colour Atlas and Textbook of Diagnostic Microbiology. Editors: Koneman E.W., Allen D.D., Dowell V.R. Jr, and Sommers H.M.
- Clinical Microbiology Procedure Handbook, Chief in editor H.D. Isenberg, Albert Einstein College of Medicine, New York, Publisher ASM (American Society for Microbiology), Washington DC.
- Jawetz, Melnick and Adelberg’s Medical Microbiology. Editors: Geo. F. Brook, Janet S. Butel & Stephen A. Morse, 21st ed 1998, Publisher Appleton & Lance, Co Stamford Connecticut.
- Manual of Clinical Microbiology. Editors: P.R. Murray, E. J. Baron, M. A. Pfaller, F. C. Tenover, and R. H. Yolken, 7th ed 2005, Publisher ASM, USA.
- Mackie and Mc Cartney Practical Medical Microbiology. Editors: J.G. Colle, A.G. Fraser, B.P. Marmion, A. Simmous, 4th ed, Publisher Churchill Living Stone, New York, Melborne, Sans Franscisco 1996.
- http://www.oxoid.com/uk/blue/prod_detail/prod_detail.asppr=CM0301&org=145&c=uk&lang=en
- https://www.bd.com/resource.aspx?IDX=8967
- https://catalog.hardydiagnostics.com/cp_prod/Content/hugo/CLEDAgar.htm
- https://www.sigmaaldrich.com/catalog/product/sial/55420lang=en®ion
- http://www.biomerieux-culturemedia.com/product/28-cled-agarA
- https://www.sciencedirect.com/science/article/abs/pii/073288939290012I

best accutane coupon
Абузоустойчивый сервер для работы с Хрумером, GSA и всевозможными скриптами!
Есть дополнительная системах скидок, читайте описание в разделе оплата
Высокоскоростной Интернет: До 1000 Мбит/с
Скорость интернет-соединения играет решающую роль в успешной работе вашего проекта. Наши VPS/VDS серверы, поддерживающие Windows и Linux, обеспечивают доступ к интернету со скоростью до 1000 Мбит/с. Это гарантирует быструю загрузку веб-страниц и высокую производительность онлайн-приложений на обеих операционных системах.
Итак, при выборе виртуального выделенного сервера VPS, обеспечьте своему проекту надежность, высокую производительность и защиту от DDoS. Получите доступ к качественной инфраструктуре с поддержкой Windows и Linux уже от 13 рублей
Абузоустойчивый сервер для работы с Хрумером, GSA и всевозможными скриптами!
Есть дополнительная системах скидок, читайте описание в разделе оплата
Высокоскоростной Интернет: До 1000 Мбит/с**
Скорость интернет-соединения – еще один важный момент для успешной работы вашего проекта. Наши VPS серверы, арендуемые под Windows и Linux, предоставляют доступ к интернету со скоростью до 1000 Мбит/с, обеспечивая быструю загрузку веб-страниц и высокую производительность онлайн-приложений на обеих операционных системах.
Абузоустойчивый сервер для работы с Хрумером, GSA и всевозможными скриптами!
Есть дополнительная системах скидок, читайте описание в разделе оплата
Виртуальные сервера (VPS/VDS) и Дедик Сервер: Оптимальное Решение для Вашего Проекта
В мире современных вычислений виртуальные сервера (VPS/VDS) и дедик сервера становятся ключевыми элементами успешного бизнеса и онлайн-проектов. Выбор оптимальной операционной системы и типа сервера являются решающими шагами в создании надежной и эффективной инфраструктуры. Наши VPS/VDS серверы Windows и Linux, доступные от 13 рублей, а также дедик серверы, предлагают целый ряд преимуществ, делая их неотъемлемыми инструментами для развития вашего проекта.
посоветуйте vps
осоветуйте vps
Абузоустойчивый сервер для работы с Хрумером и GSA и различными скриптами!
Есть дополнительная системах скидок, читайте описание в разделе оплата
Виртуальные сервера VPS/VDS и Дедик Сервер: Оптимальное Решение для Вашего Проекта
В мире современных вычислений виртуальные сервера VPS/VDS и дедик сервера становятся ключевыми элементами успешного бизнеса и онлайн-проектов. Выбор оптимальной операционной системы и типа сервера являются решающими шагами в создании надежной и эффективной инфраструктуры. Наши VPS/VDS серверы Windows и Linux, доступные от 13 рублей, а также дедик серверы, предлагают целый ряд преимуществ, делая их неотъемлемыми инструментами для развития вашего проекта.
娛樂城
2024娛樂城的創新趨勢
隨著2024年的到來,娛樂城業界正經歷著一場革命性的變遷。這一年,娛樂城不僅僅是賭博和娛樂的代名詞,更成為了科技創新和用戶體驗的集大成者。
首先,2024年的娛樂城極大地融合了最新的技術。增強現實(AR)和虛擬現實(VR)技術的引入,為玩家提供了沉浸式的賭博體驗。這種全新的遊戲方式不僅帶來視覺上的震撼,還為玩家創造了一種置身於真實賭場的感覺,而實際上他們可能只是坐在家中的沙發上。
其次,人工智能(AI)在娛樂城中的應用也達到了新高度。AI技術不僅用於增強遊戲的公平性和透明度,還在個性化玩家體驗方面發揮著重要作用。從個性化遊戲推薦到智能客服,AI的應用使得娛樂城更能滿足玩家的個別需求。
此外,線上娛樂城的安全性和隱私保護也獲得了顯著加強。隨著技術的進步,更加先進的加密技術和安全措施被用來保護玩家的資料和交易,從而確保一個安全可靠的遊戲環境。
2024年的娛樂城還強調負責任的賭博。許多平台採用了各種工具和資源來幫助玩家控制他們的賭博行為,如設置賭注限制、自我排除措施等,體現了對可持續賭博的承諾。
總之,2024年的娛樂城呈現出一個高度融合了技術、安全和負責任賭博的行業新面貌,為玩家提供了前所未有的娛樂體驗。隨著這些趨勢的持續發展,我們可以預見,娛樂城將不斷地創新和進步,為玩家帶來更多精彩和安全的娛樂選擇。
Деревянные дома под ключ
Дома АВС – Ваш уютный уголок
Мы строим не просто дома, мы создаем пространство, где каждый уголок будет наполнен комфортом и радостью жизни. Наш приоритет – не просто предоставить место для проживания, а создать настоящий дом, где вы будете чувствовать себя счастливыми и уютно.
В нашем информационном разделе “ПРОЕКТЫ” вы всегда найдете вдохновение и новые идеи для строительства вашего будущего дома. Мы постоянно работаем над тем, чтобы предложить вам самые инновационные и стильные проекты.
Мы убеждены, что основа хорошего дома – это его дизайн. Поэтому мы предоставляем услуги опытных дизайнеров-архитекторов, которые помогут вам воплотить все ваши идеи в жизнь. Наши архитекторы и персональные консультанты всегда готовы поделиться своим опытом и предложить функциональные и комфортные решения для вашего будущего дома.
Мы стремимся сделать весь процесс строительства максимально комфортным для вас. Наша команда предоставляет детализированные сметы, разрабатывает четкие этапы строительства и осуществляет контроль качества на каждом этапе.
Для тех, кто ценит экологичность и близость к природе, мы предлагаем деревянные дома премиум-класса. Используя клееный брус и оцилиндрованное бревно, мы создаем уникальные и здоровые условия для вашего проживания.
Тем, кто предпочитает надежность и многообразие форм, мы предлагаем дома из камня, блоков и кирпичной кладки.
Для практичных и ценящих свое время людей у нас есть быстровозводимые каркасные дома и эконом-класса. Эти решения обеспечат вас комфортным проживанием в кратчайшие сроки.
С Домами АВС создайте свой уютный уголок, где каждый момент жизни будет наполнен радостью и удовлетворением
ways to get money fast
Understanding the processes and protocols within a Professional Tenure Committee (PTC) is crucial for faculty members. This Frequently Asked Questions (FAQ) guide aims to address common queries related to PTC procedures, voting, and membership.
1. Why should members of the PTC fill out vote justification forms explaining their votes?
Vote justification forms provide transparency in decision-making. Members articulate their reasoning, fostering a culture of openness and ensuring that decisions are well-founded and understood by the academic community.
2. How can absentee ballots be cast?
To accommodate absentee voting, PTCs may implement secure electronic methods or designated proxy voters. This ensures that faculty members who cannot physically attend meetings can still contribute to decision-making processes.
3. How will additional members of PTCs be elected in departments with fewer than four tenured faculty members?
In smaller departments, creative solutions like rotating roles or involving faculty from related disciplines can be explored. Flexibility in election procedures ensures representation even in departments with fewer tenured faculty members.
4. Can a faculty member on OCSA or FML serve on a PTC?
Faculty members involved in other committees like the Organization of Committee on Student Affairs (OCSA) or Family and Medical Leave (FML) can serve on a PTC, but potential conflicts of interest should be carefully considered and managed.
5. Can an abstention vote be cast at a PTC meeting?
Yes, PTC members have the option to abstain from voting if they feel unable to take a stance on a particular matter. This allows for ethical decision-making and prevents uninformed voting.
6. What constitutes a positive or negative vote in PTCs?
A positive vote typically indicates approval or agreement, while a negative vote signifies disapproval or disagreement. Clear definitions and guidelines within each PTC help members interpret and cast their votes accurately.
7. What constitutes a quorum in a PTC?
A quorum, the minimum number of members required for a valid meeting, is essential for decision-making. Specific rules about quorum size are usually outlined in the PTC’s governing documents.
Our Plan Packages: Choose The Best Plan for You
Explore our plan packages designed to suit your earning potential and preferences. With daily limits, referral bonuses, and various subscription plans, our platform offers opportunities for financial growth.
Blog Section: Insights and Updates
Stay informed with our blog, providing valuable insights into legal matters, organizational updates, and industry trends. Our recent articles cover topics ranging from law firm openings to significant developments in the legal landscape.
Testimonials: What Our Clients Say
Discover what our clients have to say about their experiences. Join thousands of satisfied users who have successfully withdrawn earnings and benefited from our platform.
Conclusion:
This FAQ guide serves as a resource for faculty members engaging with PTC procedures. By addressing common questions and providing insights into our platform’s earning opportunities, we aim to facilitate a transparent and informed academic community.
side jobs from per click
Understanding the processes and protocols within a Professional Tenure Committee (PTC) is crucial for faculty members. This Frequently Asked Questions (FAQ) guide aims to address common queries related to PTC procedures, voting, and membership.
1. Why should members of the PTC fill out vote justification forms explaining their votes?
Vote justification forms provide transparency in decision-making. Members articulate their reasoning, fostering a culture of openness and ensuring that decisions are well-founded and understood by the academic community.
2. How can absentee ballots be cast?
To accommodate absentee voting, PTCs may implement secure electronic methods or designated proxy voters. This ensures that faculty members who cannot physically attend meetings can still contribute to decision-making processes.
3. How will additional members of PTCs be elected in departments with fewer than four tenured faculty members?
In smaller departments, creative solutions like rotating roles or involving faculty from related disciplines can be explored. Flexibility in election procedures ensures representation even in departments with fewer tenured faculty members.
4. Can a faculty member on OCSA or FML serve on a PTC?
Faculty members involved in other committees like the Organization of Committee on Student Affairs (OCSA) or Family and Medical Leave (FML) can serve on a PTC, but potential conflicts of interest should be carefully considered and managed.
5. Can an abstention vote be cast at a PTC meeting?
Yes, PTC members have the option to abstain from voting if they feel unable to take a stance on a particular matter. This allows for ethical decision-making and prevents uninformed voting.
6. What constitutes a positive or negative vote in PTCs?
A positive vote typically indicates approval or agreement, while a negative vote signifies disapproval or disagreement. Clear definitions and guidelines within each PTC help members interpret and cast their votes accurately.
7. What constitutes a quorum in a PTC?
A quorum, the minimum number of members required for a valid meeting, is essential for decision-making. Specific rules about quorum size are usually outlined in the PTC’s governing documents.
Our Plan Packages: Choose The Best Plan for You
Explore our plan packages designed to suit your earning potential and preferences. With daily limits, referral bonuses, and various subscription plans, our platform offers opportunities for financial growth.
Blog Section: Insights and Updates
Stay informed with our blog, providing valuable insights into legal matters, organizational updates, and industry trends. Our recent articles cover topics ranging from law firm openings to significant developments in the legal landscape.
Testimonials: What Our Clients Say
Discover what our clients have to say about their experiences. Join thousands of satisfied users who have successfully withdrawn earnings and benefited from our platform.
Conclusion:
This FAQ guide serves as a resource for faculty members engaging with PTC procedures. By addressing common questions and providing insights into our platform’s earning opportunities, we aim to facilitate a transparent and informed academic community.
일본소비세환급
일본 소비세 환급, 네오리아와 함께라면 간편하고 안전하게
일본 소비세 환급은 복잡하고 까다로운 절차로 많은 구매대행 셀러들이 어려움을 겪는 분야입니다. 네오리아는 다년간의 경험과 전문성을 바탕으로 신뢰할 수 있는 서비스를 제공하며, 일본 소비세 환급 과정을 쉽고 효율적으로 처리합니다.
1. 일본 소비세 환급의 필요성과 네오리아의 역할
네오리아는 일본 현지 법인을 설립하지 않아도 합법적인 방식으로 소비세 환급을 받을 수 있는 솔루션을 제공합니다. 이를 통해:
한국 개인사업자와 법인 사업자 모두 간편하게 환급 절차를 진행할 수 있습니다.
일본의 복잡한 서류 심사를 최소화하고, 현지 로컬 세리사와 협력하여 최적의 결과를 보장합니다.
2. 소비세 환급의 주요 특징
일본 연고가 없어도 가능: 일본에 사업자가 없더라도 네오리아는 신뢰할 수 있는 서비스를 통해 소비세 환급을 지원합니다.
서류 작성 걱정 해결: 잘못된 서류 제출로 환급이 거절될까 걱정될 필요 없습니다. 네오리아의 전문 대응팀이 모든 과정을 정밀하게 관리합니다.
현지 법인 운영자를 위한 추가 지원: 일본 내 개인사업자나 법인 운영자에게는 세무 감사와 이슈 대응까지 포함된 고급 서비스를 제공합니다.
3. 네오리아 서비스의 장점
전문성과 신뢰성: 정부로부터 인정받은 투명성과 세무 분야의 우수한 성과를 자랑합니다.
맞춤형 서포트: 다양한 사례를 통해 쌓은 경험으로 고객이 예상치 못한 어려움까지 미리 해결합니다.
로컬 업체에서 불가능한 고급 서비스: 한국인 고객을 위해 정확하고 간편한 세무회계 및 소비세 환급 서비스를 제공합니다.
4. 네오리아가 제공하는 혜택
시간 절약: 복잡한 절차와 서류 준비 과정을 전문가가 대신 처리합니다.
안심 환급: 철저한 관리와 세심한 대응으로 안전하게 환급을 받을 수 있습니다.
추가 서비스: 세무감사와 이슈 발생 시 즉각적인 지원으로 사업의 연속성을 보장합니다.
네오리아는 소비세 환급이 복잡하고 어렵다고 느껴지는 고객들에게 최적의 길잡이가 되어드립니다. 신뢰를 바탕으로 한 전문적인 서비스로, 더 이상 소비세 환급 문제로 고민하지 마세요!
wallet address checker
Overview of Digital Currency Deal Check and Regulatory Solutions
In contemporary cryptocurrency sector, ensuring transfer clarity and conformity with AML and KYC regulations is crucial. Here is an outline of leading platforms that offer services for digital asset transaction monitoring, verification, and resource security.
1. Token Metrics Platform
Overview: Token Metrics delivers digital asset analysis to evaluate possible risk dangers. This service allows individuals to review tokens ahead of purchase to avoid possibly risky assets. Attributes:
– Risk assessment.
– Ideal for investors seeking to avoid risky or fraud projects.
2. Metamask.Monitory.Center
Overview: Metamask Monitor Center enables individuals to verify their digital asset assets for questionable actions and standard compliance. Advantages:
– Verifies assets for legitimacy.
– Offers warnings about possible resource restrictions on particular exchanges.
– Delivers detailed reports after wallet sync.
3. Best Change
Overview: Bestchange.ru is a platform for monitoring and validating digital transaction deals, providing openness and deal protection. Benefits:
– Deal and wallet observation.
– Compliance screening.
– Internet interface; accommodates BTC and multiple other digital assets.
4. AML Bot
Summary: AMLCheck Bot is a holding monitor and AML tool that utilizes AI methods to identify suspicious actions. Features:
– Transfer observation and user verification.
– Accessible via internet and Telegram.
– Compatible with cryptocurrencies such as BSC, BTC, DOGE, and additional.
5. AlfaBit
Description: AlfaBit offers thorough Anti-Money Laundering (AML) services tailored for the digital currency field, helping firms and banks in ensuring regulatory adherence. Features:
– Thorough AML options and checks.
– Complies with current security and conformity guidelines.
6. AMLNode
Summary: AML Node delivers AML and identification tools for cryptocurrency firms, including transfer observing, sanctions checks, and analysis. Highlights:
– Threat evaluation options and restriction checks.
– Useful for maintaining secure company activities.
7. Btrace.AMLcrypto.io
Summary: Btrace.AMLcrypto.io specializes in fund check, offering transfer observation, compliance screenings, and support if you are a victim of theft. Benefits:
– Effective help for resource retrieval.
– Transaction monitoring and safety features.
Exclusive USDT Validation Services
Our platform also provides information on multiple sites that offer verification solutions for USDT transfers and accounts:
– **USDT TRC20 and ERC20 Validation:** Many services support thorough screenings for USDT transactions, helping in the identification of doubtful activity.
– **AML Validation for USDT:** Options are available for tracking for suspicious activities.
– **“Cleanliness” Checks for Accounts:** Validation of deal and wallet “cleanliness” is offered to identify likely dangers.
**Wrap-up**
Selecting the best tool for checking and monitoring digital currency deals is important for providing safety and compliance adherence. By viewing our evaluations, you can select the most suitable tool for deal monitoring and asset safety.
일본배대지
메인 서비스: 간편하고 효율적인 배송 및 구매 대행 서비스
1. 대행 서비스 주요 기능
메인 서비스는 고객이 한 번에 필요한 대행 서비스를 신청할 수 있도록 다양한 기능을 제공합니다.
배송대행 신청: 국내외 상품 배송을 대신 처리하며, 효율적인 시스템으로 신속한 배송을 보장합니다.
구매대행 신청: 원하는 상품을 대신 구매해주는 서비스로, 고객의 수고를 줄입니다.
엑셀 대량 등록: 대량 상품을 엑셀로 손쉽게 등록 가능하여 상업 고객의 편의성을 증대합니다.
재고 관리 신청: 창고 보관 및 재고 관리를 통해 물류 과정을 최적화합니다.
2. 고객 지원 시스템
메인 서비스는 사용자 친화적인 접근성을 제공합니다.
유저 가이드: 대행 서비스를 더욱 합리적으로 사용할 수 있도록 세부 안내서를 제공합니다.
운송장 조회: 일본 사가와 등 주요 운송사의 추적 시스템과 연동하여 운송 상황을 실시간으로 확인 가능합니다.
3. 비용 안내와 부가 서비스
비용 계산기: 예상되는 비용을 간편하게 계산해 예산 관리를 돕습니다.
부가 서비스: 교환 및 반품, 폐기 및 검역 지원 등 추가적인 편의 서비스를 제공합니다.
출항 스케줄 확인: 해외 배송의 경우 출항 일정을 사전에 확인 가능하여 배송 계획을 세울 수 있습니다.
4. 공지사항
기본 검수 공지
무료 검수 서비스로 고객의 부담을 줄이며, 보다 철저한 검수가 필요한 경우 유료 정밀 검수 서비스를 권장합니다.
수출허가서 발급 안내
항공과 해운 수출 건에 대한 허가서를 효율적으로 발급받는 방법을 상세히 안내하며, 고객의 요청에 따라 이메일로 전달됩니다.
노데이터 처리 안내
운송장 번호 없는 주문에 대한 새로운 처리 방안을 도입하여, 노데이터 발생 시 관리비가 부과되지만 서비스 품질을 개선합니다.
5. 고객과의 소통
카카오톡 상담: 실시간 상담을 통해 고객의 궁금증을 해결합니다.
공지사항 알림: 서비스 이용 중 필수 정보를 지속적으로 업데이트합니다.
메인 서비스는 고객 만족을 최우선으로 하며, 지속적인 개선과 세심한 관리를 통해 최상의 경험을 제공합니다.
일본배대지
메인 서비스: 간편하고 효율적인 배송 및 구매 대행 서비스
1. 대행 서비스 주요 기능
메인 서비스는 고객이 한 번에 필요한 대행 서비스를 신청할 수 있도록 다양한 기능을 제공합니다.
배송대행 신청: 국내외 상품 배송을 대신 처리하며, 효율적인 시스템으로 신속한 배송을 보장합니다.
구매대행 신청: 원하는 상품을 대신 구매해주는 서비스로, 고객의 수고를 줄입니다.
엑셀 대량 등록: 대량 상품을 엑셀로 손쉽게 등록 가능하여 상업 고객의 편의성을 증대합니다.
재고 관리 신청: 창고 보관 및 재고 관리를 통해 물류 과정을 최적화합니다.
2. 고객 지원 시스템
메인 서비스는 사용자 친화적인 접근성을 제공합니다.
유저 가이드: 대행 서비스를 더욱 합리적으로 사용할 수 있도록 세부 안내서를 제공합니다.
운송장 조회: 일본 사가와 등 주요 운송사의 추적 시스템과 연동하여 운송 상황을 실시간으로 확인 가능합니다.
3. 비용 안내와 부가 서비스
비용 계산기: 예상되는 비용을 간편하게 계산해 예산 관리를 돕습니다.
부가 서비스: 교환 및 반품, 폐기 및 검역 지원 등 추가적인 편의 서비스를 제공합니다.
출항 스케줄 확인: 해외 배송의 경우 출항 일정을 사전에 확인 가능하여 배송 계획을 세울 수 있습니다.
4. 공지사항
기본 검수 공지
무료 검수 서비스로 고객의 부담을 줄이며, 보다 철저한 검수가 필요한 경우 유료 정밀 검수 서비스를 권장합니다.
수출허가서 발급 안내
항공과 해운 수출 건에 대한 허가서를 효율적으로 발급받는 방법을 상세히 안내하며, 고객의 요청에 따라 이메일로 전달됩니다.
노데이터 처리 안내
운송장 번호 없는 주문에 대한 새로운 처리 방안을 도입하여, 노데이터 발생 시 관리비가 부과되지만 서비스 품질을 개선합니다.
5. 고객과의 소통
카카오톡 상담: 실시간 상담을 통해 고객의 궁금증을 해결합니다.
공지사항 알림: 서비스 이용 중 필수 정보를 지속적으로 업데이트합니다.
메인 서비스는 고객 만족을 최우선으로 하며, 지속적인 개선과 세심한 관리를 통해 최상의 경험을 제공합니다.
일본 소비세 환급, 네오리아와 함께라면 간편하고 안전하게
일본 소비세 환급은 복잡하고 까다로운 절차로 많은 구매대행 셀러들이 어려움을 겪는 분야입니다. 네오리아는 다년간의 경험과 전문성을 바탕으로 신뢰할 수 있는 서비스를 제공하며, 일본 소비세 환급 과정을 쉽고 효율적으로 처리합니다.
1. 일본 소비세 환급의 필요성과 네오리아의 역할
네오리아는 일본 현지 법인을 설립하지 않아도 합법적인 방식으로 소비세 환급을 받을 수 있는 솔루션을 제공합니다. 이를 통해:
한국 개인사업자와 법인 사업자 모두 간편하게 환급 절차를 진행할 수 있습니다.
일본의 복잡한 서류 심사를 최소화하고, 현지 로컬 세리사와 협력하여 최적의 결과를 보장합니다.
2. 소비세 환급의 주요 특징
일본 연고가 없어도 가능: 일본에 사업자가 없더라도 네오리아는 신뢰할 수 있는 서비스를 통해 소비세 환급을 지원합니다.
서류 작성 걱정 해결: 잘못된 서류 제출로 환급이 거절될까 걱정될 필요 없습니다. 네오리아의 전문 대응팀이 모든 과정을 정밀하게 관리합니다.
현지 법인 운영자를 위한 추가 지원: 일본 내 개인사업자나 법인 운영자에게는 세무 감사와 이슈 대응까지 포함된 고급 서비스를 제공합니다.
3. 네오리아 서비스의 장점
전문성과 신뢰성: 정부로부터 인정받은 투명성과 세무 분야의 우수한 성과를 자랑합니다.
맞춤형 서포트: 다양한 사례를 통해 쌓은 경험으로 고객이 예상치 못한 어려움까지 미리 해결합니다.
로컬 업체에서 불가능한 고급 서비스: 한국인 고객을 위해 정확하고 간편한 세무회계 및 소비세 환급 서비스를 제공합니다.
4. 네오리아가 제공하는 혜택
시간 절약: 복잡한 절차와 서류 준비 과정을 전문가가 대신 처리합니다.
안심 환급: 철저한 관리와 세심한 대응으로 안전하게 환급을 받을 수 있습니다.
추가 서비스: 세무감사와 이슈 발생 시 즉각적인 지원으로 사업의 연속성을 보장합니다.
네오리아는 소비세 환급이 복잡하고 어렵다고 느껴지는 고객들에게 최적의 길잡이가 되어드립니다. 신뢰를 바탕으로 한 전문적인 서비스로, 더 이상 소비세 환급 문제로 고민하지 마세요!
Solutions for Verifying USDT for Embargoes and Operation Purity: Money Laundering Prevention Strategies
In the contemporary domain of crypto assets, where quick transactions and obscurity are becoming the usual case, monitoring the lawfulness and clarity of processes is crucial. In view of increased administrative scrutiny over illicit finance and financing of terrorism, the need for robust instruments to authenticate transactions has become a major concern for digital asset users. In this text, we will explore existing solutions for assessing USDT for embargoes and transaction clarity.
What is AML?
AML strategies refer to a collection of supervisory protocols aimed at curtailing and identifying dirty money activities. With the increase of digital asset usage, AML standards have become especially crucial, allowing participants to manage digital currencies reliably while mitigating risks associated with embargoes.
USDT, as the most well-known stablecoin, is broadly used in different transactions across the globe. Yet, using USDT can present several hazards, especially if your funds may tie to opaque or criminal activities. To reduce these risks, it’s crucial to take make use of offerings that check USDT for sanctions.
Available Services
1. Address Authentication: Using customized tools, you can verify a specific USDT address for any connections to sanction registries. This aids identify potential associations to illicit conduct.
2. Deal Engagement Evaluation: Some platforms provide scrutiny of operation records, crucial for measuring the clarity of fund flows and detecting potentially risky activities.
3. Surveillance Services: Specialized monitoring services allow you to follow all exchanges related to your address, facilitating you to quickly identify concerning activities.
4. Threat Records: Certain platforms provide detailed concern reports, which can be valuable for participants looking to validate the integrity of their holdings.
Regardless of whether you are managing a large capital or performing small transactions, following to AML norms supports steer clear of legal repercussions. Employing USDT verification solutions not only protects you from economic setbacks but also helps to creating a safe environment for all economic stakeholders.
Conclusion
Checking USDT for embargoes and deal cleanliness is becoming a required measure for anyone enthusiastic to remain within the legal framework and support high benchmarks of openness in the digital asset industry. By collaborating with dependable solutions, you not only safeguard your resources but also contribute to the common goal in combating illicit finance and terror financing activities.
If you are willing to start leveraging these services, investigate the accessible options and choose the solution that most suits your needs. Be aware, information is your advantage, and timely operation validation can rescue you from countless problems in the coming times.
hi!,I like your writing very much! percentage we be in contact extra about your post on AOL? I require a specialist in this house to unravel my problem. Maybe that is you! Having a look forward to peer you.