Introduction of Cryptosporidium
Table of Contents
Cryptosporidium is a coccidian parasite that causes diarrhea called Cryptosporidiosis. It was discovered by medical parasitologist, Ernest Edward in 1907. Human infection caused by Cryptosporidium parvum was seen in 1976. The clinically important species of this genus are C. parvum, C. mammals, and C. muris.
Scientific Classification of C. parvum
| Kingdom | Kingdom |
| Superphylum | Alveolata |
| Phylum | Apicomplexa |
| Class | Conoidasida |
| Order | Eucoccidiorida |
| Suborder | Eimeriorina |
| Family | Cryptosporidiidae |
| Genus | Cryptosporidium |
| Species | C. parvum |
Geographical Distribution
This parasite is cosmopolitan and distributed worldwide. It is found in India; China Nepal and Southeast Asia. Habitat-C. parvum is intracellular, found within the micro-villous region of epithelial cells of the small intestine. It has also been found, but less frequently, in the stomach, large intestine, and even lungs.
Morphology of Cryptosporidium parvum
Oocyst is oval or spherical of size 4-5µm in diameter. It has a colorless, transparent shell. The oocyst wall of C. parum is composed of an electron-lucent, middle zone surrounded by two electron-dense layers and has four crescent-shaped sporozoites. Oocyst does not stain with iodine and it is acid-fast. The anterior end of the sporozoites is pointed and the posterior end, which contains a prominent nucleus, is rounded.
Life Cycle of Cryptosporidium parvum
C. parvum undergoes both sexual and asexual multiplication in a single host; cattle, cat, or dog. Sporulated oocysts containing 4 sporozoites are excreted by the infected host through feces. Transmission of cysts of C. parvum and C. hominis occurs mainly through contaminated water and feces of infected animals. Following ingestion by a suitable host, then excystation occurs. The sporozoites are released and parasitize epithelial cells of the gastrointestinal tract. In these cells, the parasites undergo sexual multiplication (schizogony). Then sexual multiplication (gametogony) produces microgametes (male) and macrogamonts (female). Upon fertilization of the macrogamonts by the microgametes, oocysts develop that sporulate in the infected host. Two types of oocysts are produced, the thick–walled, which is commonly excreted from the host, and the thin-walled oocyst, which is primarily in autoinfection.
Pathogenicity of Cryptosporidium parvum
Infection with Cryptosporidium results in a wide range of manifestations, from asymptomatic infection
too severe, life-threatening illness. Water diarrhea is the most frequent symptom and can be accompanied by dehydration, weight loss, abdominal pain and cramps, fever, nausea, and vomiting. In immunocompetent hosts, symptoms are usually short (1 to 2 weeks). Infection in healthy, immunocompetent persons is self-limited. The disease can be chronic and more severe in immunocompromised patients, particularly with AIDS especially those with CD4 counts less than 200/µl. The small intestine is the site most commonly affected, symptomatic Cryptosporidium infections have
also been founded in other organs including the gastrointestinal (GI) tract, lung, and conjunctiva.
Laboratory Diagnosis of Cryptosporidium parvum
Diagnosis can be made by stool or fecal examination. Acid–fast/Ziehl-Neelsen staining methods, with or without stool concentration, are most frequently used in clinical laboratories. For the greatest sensitivity and specificity, immunofluorescence microscopy is the method of choice (followed closely by enzyme
immunoassays). A molecular method is mainly a research tool.
- Direct Method-Specimen processing: Stool specimens preserved in 10% buffered or suspended in a storage medium composed of aqueous potassium dichromate (25% w/v, final concentration). Multiply stool samples should be tested before a negative diagnostic interpretation is reported.
- Wet Mount-Wet mount examination with iodine is used mainly for screening and is especially useful with specimens containing moderate to high numbers of oocysts. However, it is should be combined with a more sensitive confirmatory stain or assay. Fresh or concentrated fecal specimens can be examined, using either conventional bright light, phase contrast, or differential interference contrast microscopy.
- Sedimentation methods- Stool samples should be concentrated prior to microscopic examination. Formalin–ethyl acetate sedimentation is the recommended stool concentration method for clinical laboratories. Sedimentation methods are generally performed using low-speed centrifugation. Given their small mass, Cryptosporidium oocysts may become trapped in the ether or ethyl acetate plug and fail to sediment properly. Increased centrifugation speed or time (500×g, 10 minutes) may be warranted when attempting to recover cryptosporidial oocysts.
Stained Smears
- Giemsa stain: Traditional stains, Giemsa is of limited value. They do not differentiate between oocysts, similarly sized fecal yeasts, and other debris.
- Modified Acid -Fast staining technique: Modified Acid fast staining or Kinyoun’s staining technique is a simple and effective method. The acid-fast staining technique has been modified and improved including hot and cold modified acid–fast stains, incorporation of dimethyl sulfoxide (DMSO), and incorporation of the detergent tergitol. Oocysts are acid-fast and stained with a faint pink-red color. Oocysts stain orange-red with safranin.
- Biopsy: Duodenal and jejunal biopsy
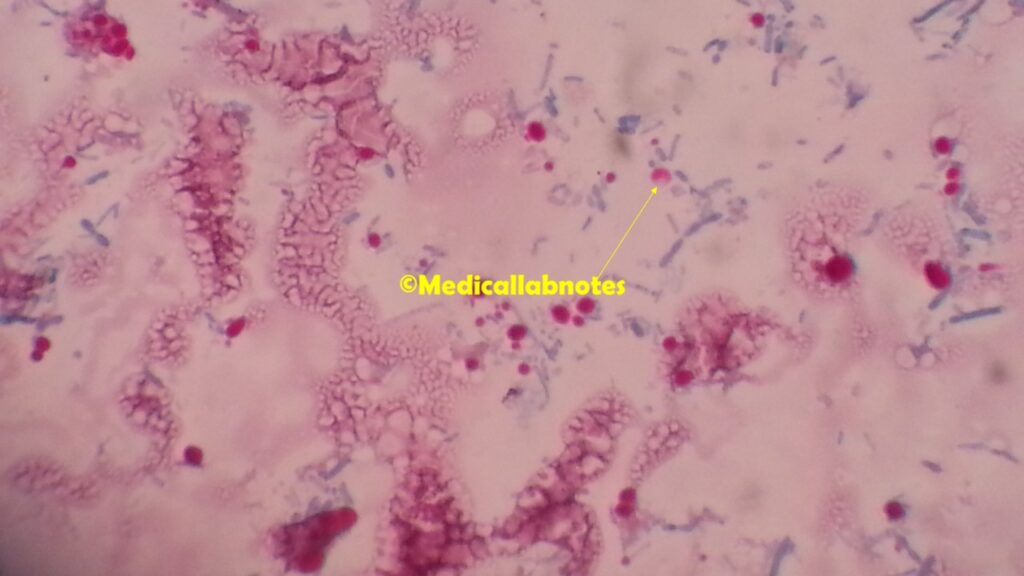
Microscopic detection of oocysts by the following methods-
- Alternate bright–field stains
- Hot safranin – methylene blue stain,
- Modified Kohn’s stain
- Aniline- carbol – methyl violet and
- Tartrazine
- Negative stains
- Fluorescent stains
- Including auramine O,
- Auramine – rhodamine,
- Auramine- carbol – fuchsin, acridine orange and
- Mepacrine (vital stain)
- Enzyme immunoassays (EIA) have been introduced for the detection of crystosporidial antigens in stool samples. These tests are superior to conventional microscopic examination (especially acid-fast staining methods) and show a good correlation with monoclonal antibody–based immunofluorescence assays.
- Immunofluorescence microscopy for detection of oocysts: This method offers increased sensitivity and specificity compared to staining techniques. It has found widespread application in research and clinical laboratories as well as for monitoring oocyst presence in environmental samples. The assays generally work well with fresh or preserved stools (formalin, potassium dichromate).
Antibody detection: There are currently no commercially available serologic assays for the detection of Cryptosporidium-specific antibodies. However, immunoblots for detecting the 17 and 27 kg kDa sporozoite antigens associated with recent infection may be useful for epidemiologic investigation.
Molecular diagnosis: Analysis of a PCR diagnostic test for detection of C. parvum DNA. A PCR test can be done by using primers.
Prevention, Control, and Treatment of Cryptosporidiosis
- Electrolyte replacement: Rapid loss of fluids because diarrhea can be managed by fluid and electrolyte replacement.
- Immunocompromised hosts and those in poor health are at the highest risk for severe illness. Co-trimoxazole appears to be effective in the management of serious infections.
- Nitazoxanide gas provided some encouraging results in the management of cryptosporidial diarrhea in immunocompetent patients and immunocompetent children under 12 years old.
- For people with AIDS, anti-retroviral therapy, which improves immune status, will also reduce oocyst excretion and decrease diarrhea associated with cryptosporidiosis.
Keynotes
Cryptosporidium parvum
Size of parasite: 4.5 -5.5 µm
Site in host: small intestine, micro–villa
Mode of infection
Portal of entry: Mouth, ingestion
Causative agent: Oocyst
Medium: Contaminated food and water
Clinical symptoms: Watery diarrhea
Disease: Cryptosporidiosis
Further Reading
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2716703/
- https://en.wikipedia.org/wiki/Cryptosporidium
- Isenberg clinical microbiology procedures Handbook
2nd edition. Vol. 2 - Markell and Voge’s medical parasitology
9th edition. - Parasitology: 12th edition
By K. D. Chatterjee - District laboratory practice in Tropical countries –Part-I.
By Monica Chesbrough. - Atlas of Medical Helminthology and protozoology -4th edn -P.L. Chiodini, A.H. Moody, D.W. Manser
- Medical Parasitology by Abhay R. Satoskar, Gary L. Simon, Peter J. Hotez and Moriya Tsuji
