Introduction
Table of Contents
The Formal Ether Concentration Technique is a laboratory method used to concentrate parasitic organisms, such as eggs, larvae, or cysts, from a fecal sample. It is particularly useful for the detection of intestinal parasites that may be present in low numbers and are difficult to identify using direct smear microscopy alone.
The technique involves using a concentrated solution of formalin and ether to separate the parasitic organisms from the fecal material. The formalin acts as a preservative, while the ether serves as a solvent to dissolve fecal debris and lipids, leaving behind the concentrated parasite material.
The Formal Ether Concentration Technique is commonly employed in parasitology laboratories for the diagnosis of various gastrointestinal parasites, including helminths (such as Ascaris, Trichuris, and hookworms) and protozoa (such as Giardia and Entamoeba species).
The concentrated parasite material obtained through this technique can then be examined under a microscope. It allows for better visualization and identification of the parasites. This method enhances the sensitivity of parasite detection compared to direct smear microscopy alone. It helps to remove interfering fecal debris and concentrate the target organisms.
It is important to note that this technique requires proper handling of formalin and ether, as they can be hazardous if not used correctly. Adequate laboratory safety precautions should be followed during the procedure to ensure the well-being of laboratory personnel.
The Formal Ether Concentration Technique is a valuable tool in the diagnosis of intestinal parasites, as it helps improve the detection sensitivity and accuracy of parasitological examinations.
Principle
The principle of the Formal Ether Concentration Technique is based on the differential solubility of parasitic organisms, specifically their eggs, larvae, or cysts, in a concentrated formalin-ether solution compared to fecal debris and other substances present in the sample.
The technique works on the principle that formalin acts as a preservative and fixes the parasites, while ether serves as a solvent that dissolves fecal debris and lipids. This solvent action helps to separate the parasite material from the bulk of the fecal material.
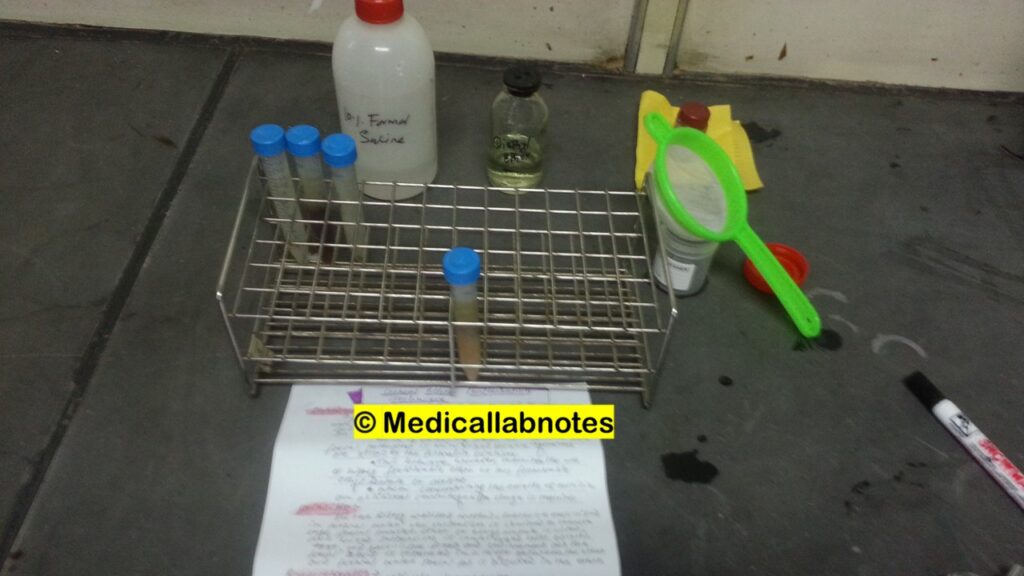
Test Requirements
The Formal Ether Concentration Technique requires specific test requirements and materials to perform the procedure effectively. Here are the typical requirements for conducting the Formal Ether Concentration Technique:
- Fecal Sample: A fresh fecal sample is needed for analysis. It is preferable to collect the sample in a clean, dry container and avoid any contamination or contact with urine or water.
- 10% Formalin Solution: A 10% formalin solution is required as a preservative for fixing the parasites and preventing their degradation. This solution can be prepared by diluting formalin (37-40% formaldehyde) with distilled water in the appropriate ratio.
- Centrifuge Tubes: Suitable centrifuge tubes are required to hold the sample and facilitate the centrifugation process. These tubes should be capable of withstanding high centrifugal forces.
- Centrifuge: A centrifuge is necessary to spin the centrifuge tubes at a high speed. It should be capable of reaching sufficient rotational speed to separate the parasite material from the rest of the sample.
- Gauze or Mesh: A piece of gauze or mesh is used to filter the fecal sample, removing larger debris and solid particles. It helps in obtaining a cleaner filtrate for further processing.
- Ether: Ether is a solvent that is added to the concentrated sample to dissolve fecal debris and lipids. It aids in the separation of the dissolved debris from the parasite material.
- Microscope: A light microscope is required for the examination of the concentrated sediment after the Formal Ether Concentration Technique. It should have appropriate magnification and illumination settings for visualizing the parasites.
- Staining Reagents (Optional): Depending on the specific parasites being targeted, staining reagents. e.g. iodine or modified acid-fast stains may be needed to enhance the visibility of the parasites during microscopic examination.
- Personal Protective Equipment (PPE): It is important to wear appropriate PPE, including gloves, laboratory coat or gown, and safety goggles, to ensure personal safety and minimize the risk of exposure to formalin and other hazardous materials.
It is worth noting that specific laboratory protocols and procedures may vary. Therefore, it is important to refer to standard operating procedures and guidelines. It provides by the laboratory or consult appropriate references for the accurate and detailed requirements specific to the Formal Ether Concentration Technique in your laboratory setting.
Test Procedure
The following is a general procedure for performing the Formal Ether Concentration Technique:
- First, wear gloves when handling stool specimens.
- In a suitable container, thoroughly mix a portion of stool specimen approximately 1 ml or the size of a walnut into 10 ml of normal saline. Mix thoroughly with the help of a vortex.
- Filter the emulsion through fine mesh gauze or alternatively wire sieve into a conical centrifuge tube as shown above picture.
- Centrifuge the suspension at 2000 rpm for 10 minutes. Note: The suspension should yield about 0.75 ml of sediment for fresh specimens and 0.5 ml for formalized feces.
- Decant the supernatant and wash the sediment with 10 ml of saline solution. Centrifuge again and repeat washing until the supernatant is clear.
- After the last wash, decant the supernatant and add 10 ml of 10% formalin to the sediment. Mix and let stand for 5 minutes to effect fixation.
- Add 1 to 2 ml of ethyl acetate, Stopper the tube, and shake vigorously.
- Centrifuge at 1500 rpm for 10 minutes. Four layers should result as a top layer of ethyl acetate, plug of debris, layer of formalin, and sediment respectively.
- Free the plug of debris from the side of the tube by ringing with an applicator stick. Carefully decant the top three layers.
- Mix the remaining sediment with a pipette
- Transfer a small portion of the resuspended sediment onto a microscope slide using a pipette or applicator stick. Optional staining techniques, such as iodine or modified acid-fast stains, can be applied at this stage to enhance the visibility of the parasites.
- Place a coverslip over the slide and examine it under a microscope using appropriate magnification. Look for the presence of parasite eggs, larvae, cysts, or other structures.
Uses
The Formal Ether Concentration Technique is widely used in parasitology laboratories for the diagnosis of various gastrointestinal parasites. Here are some of the key uses of the Formal Ether Concentration Technique:
- Detection of Intestinal Parasites: The technique is primarily employed for the detection of intestinal parasites, including both helminths (e.g., Ascaris, Trichuris, hookworms) and protozoa (e.g., Giardia, Entamoeba species). It helps in concentrating and visualizing the parasite stages (eggs, larvae, cysts) present in fecal samples, enhancing the sensitivity of detection.
- Improved Sensitivity: The Formal Ether Concentration Technique improves the sensitivity of parasite detection compared to direct smear microscopy alone. It allows for the concentration of parasites that may be present in low numbers, increasing the chances of their identification.
- Differentiation of Parasites: By concentrating the parasite material, the technique aids in the differentiation of various parasitic species. This is particularly valuable when multiple parasite species are suspected or when distinguishing between similar-looking parasite stages is necessary.
- Enhanced Visualization: The technique helps remove interfering fecal debris and lipids, resulting in a cleaner sample for microscopic examination. This enhances the visibility of the parasites, making it easier to identify their characteristic morphological features.
- Zoonotic Parasite Identification: Zoonotic parasites, which can be transmitted between animals and humans, can also be detected using the Formal Ether Concentration Technique. Examples include parasites like Cryptosporidium and Toxoplasma, which pose risks to both human and animal health.
- Research and Surveillance: The technique plays a vital role in research studies and surveillance programs focused on studying the prevalence, distribution, and epidemiology of gastrointestinal parasites. It helps in obtaining accurate data on parasite occurrence and identifying potential public health risks.
Keynotes
Principle: The technique utilizes the differential solubility of parasites in a concentrated formalin-ether solution. Formalin acts as a preservative, fixing the parasites, while ether dissolves fecal debris and lipids, leaving behind the concentrated parasite material.Sample Preparation: A small amount of fecal sample is mixed with 10% formalin solution and filtered to remove larger debris. The filtrate is then subjected to centrifugation to concentrate the parasite material.Ether Addition: After removing the supernatant, ether is added to the concentrated sediment, and the tube is vigorously mixed. Ether dissolves fecal debris and lipids, facilitating the separation of dissolved debris from the parasite material.
Sediment Examination: The upper layer, containing dissolved debris and ether, is poured off, and the sediment is resuspended in saline or water. A portion of the resuspended sediment is then examined under a microscope for the presence of parasite stages.Enhanced Sensitivity: The Formal Ether Concentration Technique improves the sensitivity of parasite detection compared to direct smear microscopy alone. It allows for the concentration of parasites, enhancing their visibility and increasing the chances of detection. Identification of Various Parasites: The technique is useful for identifying a wide range of gastrointestinal parasites, including helminths (worms) and protozoa. It aids in the visualization and differentiation of parasite stages, contributing to accurate species identification.Laboratory Safety: The use of formalin and ether requires adherence to proper safety protocols. It is crucial to work in a well-ventilated area or under a fume hood, wear appropriate personal protective equipment (PPE), and follow the laboratory’s safety guidelines.
Complementary Techniques: The Formal Ether Concentration Technique is often used in conjunction with other diagnostic methods, such as direct smear microscopy, to enhance parasite detection. Different techniques may be employed depending on the specific parasite and the laboratory’s capabilities.Diagnostic Applications: The technique finds utility in clinical diagnostics, research, and surveillance of gastrointestinal parasites. It aids in identifying and studying parasites of medical and veterinary importance, contributing to effective patient management and public health interventions.Interpretation: Proper training and expertise are required for accurate interpretation of the microscopic findings. Knowledge of parasite morphology and characteristic features is essential for distinguishing between different parasite species and stages.
Remember that specific laboratory protocols may vary, so it is important to follow the established guidelines and consult appropriate references for detailed instructions and modifications specific to your laboratory setting.
Further Readings
- Cheesbrough, M. (2006). District Laboratory Practice in Tropical Countries: Part 1 (2nd ed.). Cambridge University Press.
- Garcia, L. S. (2016). Diagnostic Medical Parasitology (6th ed.). ASM Press.
- Garcia, L. S., & Shimizu, R. Y. (2009). Evaluation of intestinal parasites in stool specimens: Enhanced methods and further validation of new techniques. Journal of Clinical Microbiology, 47(9), 2965-2977.
- Haque, R., Huston, C. D., Hughes, M., Houpt, E., & Petri Jr, W. A. (2003). Amebiasis. New England Journal of Medicine, 348(16), 1565-1573.
- Procop, G. W., & Church, D. L. (2017). Laboratory procedures for diagnosis of parasitic infections. Clinical Microbiology Reviews, 30(1), 67-100.
- WHO. (1991). Basic Laboratory Methods in Medical Parasitology. World Health Organization.
- Zajac, A. M., & Conboy, G. A. (2012). Veterinary Clinical Parasitology (8th ed.). John Wiley & Sons.

Hi my friend! I wish to say that this post is amazing, great written and include almost all important infos.I would like to peer more posts like this .
Hi there, just became aware of your blog through Google, and found that it is really informative. I’m gonna watch out for brussels. I will appreciate if you continue this in future. Many people will be benefited from your writing. Cheers!
No matter if some one searches for his essential thing, therefore he/she desires to be available that in detail, therefore that thing is maintained over here.
Hey, thanks for the article post. Really Great.
I wanted to thank you for this wonderful read!! I definitely loved every bit ofit. I’ve got you saved as a favorite to lookat new stuff you post…
It’s actually a nice and useful piece of info. I’m satisfied that you just sharedthos helpful information with us. Please stay us up tto date likethis. Thanks for sharing.
Hey there! This is my first visit to your blog! We are a collection of volunteers andstarting a new initiative in a community in the same niche.Your blog provided us valuable information to workon. You have done a outstanding job!
I value the article post.Really thank you! Will read on…
I appreciate you sharing this blog article.Really thank you! Will read on…
Asking questions are truly nice thing if you are not understanding anything totally, but this article gives fastidious understanding even.
This is a very beneficial little bit of facts aplikasi prediksi saham terkini. Now i’m satisfied that you just provided this beneficial information with us. Be sure to stop us educated in this way. Many thanks discussing.
A big thank you for your article.Thanks Again. Really Great.
Very informative blog post.Really looking forward to read more. Really Cool.
I value the blog post. Cool.
Thanks for the blog article.Really looking forward to read more. Really Cool.
Very informative blog post.Really thank you! Really Great.
I cannot thank you enough for the blog post.Really thank you! Keep writing.
Wow, great blog article.Really thank you! Fantastic.
Hello friends, how is all, and what you wish for to say regarding this post,in my view its in fact remarkable in support of me.