Introduction
Table of Contents
SARS-CoV-2 antigen tests, also known as rapid antigen tests or rapid diagnostic tests (RDTs), are diagnostic tests designed to detect specific proteins or antigens from the SARS-CoV-2 virus, which is responsible for the COVID-19 disease. These tests are used to quickly identify active COVID-19 infections in individuals.
The SARS-CoV-2 antigen test works by detecting the presence of viral proteins in respiratory specimens, typically collected from the nose or throat using a swab. The sample is then processed using a test kit that contains antibodies specific to SARS-CoV-2 antigens. If the viral antigens are present in the sample, they bind to the antibodies, leading to a visual or automated indication of a positive test result.
Antigen tests are generally designed to provide rapid results, often within minutes, making them valuable tools for point-of-care testing in various settings, including healthcare facilities, clinics, and community testing centers. They are particularly useful for screening large numbers of individuals quickly and identifying individuals who may be contagious and require further confirmatory testing or isolation.
It’s important to note that antigen tests have a slightly lower sensitivity compared to molecular tests like PCR (polymerase chain reaction), which are considered the gold standard for COVID-19 diagnosis. Antigen tests may have a higher chance of producing false-negative results, especially during the early stages of infection or in individuals with lower viral loads. Therefore, if the antigen test result is negative but there is a high suspicion of COVID-19, a molecular test may be recommended to confirm the result.
The use of SARS-CoV-2 antigen tests has been instrumental in expanding access to testing, enabling rapid identification and isolation of infected individuals, and facilitating timely public health interventions. However, it’s important to follow the guidance and recommendations of public health authorities regarding the appropriate use and interpretation of antigen tests in different settings and populations.
Principle
The principle of SARS-CoV-2 antigen tests involves the detection of specific viral proteins or antigens from respiratory specimens to diagnose active COVID-19 infections. These tests are designed to provide rapid results, often within minutes, making them valuable tools for point-of-care testing.
The specific steps and components of SARS-CoV-2 antigen tests may vary depending on the test manufacturer. However, the general principle involves the following steps:
- Sample collection: A respiratory specimen, typically obtained from the nose or throat using a swab, is collected from the individual suspected of having COVID-19. The swab is inserted into a collection tube containing a transport medium.
- Sample preparation: The collected specimen is mixed with a buffer or reagent to prepare the sample for testing. This step may involve vortexing or shaking the sample to ensure proper mixing.
- Test application: A portion of the prepared sample is then applied to a test device, such as a lateral flow immunoassay strip or a microfluidic cartridge. The device contains specific antibodies that are designed to detect SARS-CoV-2 viral antigens.
- Antigen-antibody reaction: If SARS-CoV-2 viral antigens are present in the sample, they bind to the antibodies immobilized on the test device. The antibodies are typically specific to viral proteins, such as the nucleocapsid protein or the spike protein of the virus.
- Signal generation: Depending on the test design, the antigen-antibody reaction may generate a visible signal, such as a colored line, indicating a positive result. Some tests may also utilize fluorescence or chemiluminescence technology for signal detection. The absence of a visible signal indicates a negative result.
- Interpretation of results: The test results are interpreted based on the appearance or absence of the visible signal according to the manufacturer’s instructions. It’s important to follow the specific guidelines provided with the test kit for result interpretation.
Test Requirements
The specific test requirements for SARS-CoV-2 antigen tests can vary depending on the test manufacturer and the regulatory guidelines of different countries or health authorities. However, here are some common requirements and considerations for conducting SARS-CoV-2 antigen testing:
- Sample collection: SARS-CoV-2 antigen tests typically require a respiratory specimen, such as a nasal or throat swab, to be collected from the individual being tested. The specimen should be collected using appropriate collection devices and techniques specified by the test manufacturer or regulatory guidelines.
- Test kit: SARS-CoV-2 antigen tests require the use of a specific test kit or assay provided by the manufacturer. The test kit usually contains all the necessary components, including the test device, buffers, reagents, and instructions for use. It’s important to follow the instructions provided with the test kit for accurate testing.
- Testing environment: SARS-CoV-2 antigen tests can be performed in various settings, including healthcare facilities, clinics, or community testing centers. The testing environment should be clean and well-equipped with necessary supplies, including personal protective equipment (PPE), appropriate waste disposal systems, and facilities for safe handling of specimens.
- Personnel training: Adequate training is crucial for healthcare professionals or individuals performing the antigen test. They should be trained on proper sample collection techniques, test kit handling, and result interpretation. Following the manufacturer’s instructions and any additional training or guidelines provided by local health authorities is essential.
- Quality control: Quality control measures should be implemented to ensure the accuracy and reliability of the antigen test results. This may involve running positive and negative controls provided by the manufacturer alongside patient samples to verify the performance of the test and the test kit.
- Test interpretation: Clear guidelines for result interpretation should be provided by the manufacturer. The appearance or absence of a visible signal, such as a colored line, indicates a positive or negative result, respectively. It’s important to follow the specified time frame for result interpretation, as well as any additional guidance provided by the manufacturer or local health authorities.
- Reporting and data management: Test results should be accurately recorded and reported according to local guidelines and requirements. This may involve proper documentation of patient information, test results, and any relevant metadata, such as date and time of testing. Data management protocols should be in place to ensure confidentiality and secure storage of test results.
Procedure
The procedure for conducting a SARS-CoV-2 antigen test can vary depending on the specific test kit and manufacturer instructions. However, here is a general outline of the procedure:
- Pre-test preparation:
- Gather all necessary materials and ensure they are within the specified expiration dates.
- Review the manufacturer’s instructions for the specific test kit being used.
- Put on appropriate personal protective equipment (PPE), such as gloves, a mask, and a gown, to ensure safety during the testing process.
- Set up a clean and well-lit testing area.
- Sample collection:
- Collect a respiratory specimen from the individual being tested. The most common method is by using a nasal or throat swab.
- Follow the manufacturer’s instructions for proper collection techniques and specimen transport.
- Sample preparation:
- Place the collected swab into a tube containing a buffer or reagent provided with the test kit.
- Follow the manufacturer’s instructions for sample preparation, which may involve vortexing, shaking, or swirling the specimen in the tube to ensure proper mixing.
- Test application:
- Open the test kit package and remove the test device.
- Using a dropper or provided transfer device, add a specific amount of the prepared sample to the test device. The sample is usually added to a designated well or area on the test device.
- Incubation:
- Allow the test device to incubate for the specified time period, usually a few minutes. This allows the antigen-antibody reaction to occur.
- Result interpretation:
- After the incubation period, read the test results according to the manufacturer’s instructions.
- Look for any visible signals, such as the appearance of colored lines or other indicators on the test device.
- Interpret the results as positive, negative, or invalid, based on the presence or absence of the specific signals.
- Result documentation and reporting:
- Record the test results accurately and legibly.
- Follow local guidelines and reporting requirements for documenting and reporting test results, including patient information, test date and time, and any other required data.
- Post-test procedures:
- Dispose of used test materials, such as swabs and tubes, according to local regulations and guidelines.
- Clean and disinfect the testing area and any equipment used during the testing process.
- Properly store and maintain records of the test results, as required.
Result-Interpreation
The interpretation of SARS-CoV-2 antigen test results is typically based on the presence or absence of visible signals, such as colored lines or other indicators, on the test device. The specific interpretation criteria may vary depending on the manufacturer’s instructions and the test kit being used. However, here are some general guidelines:
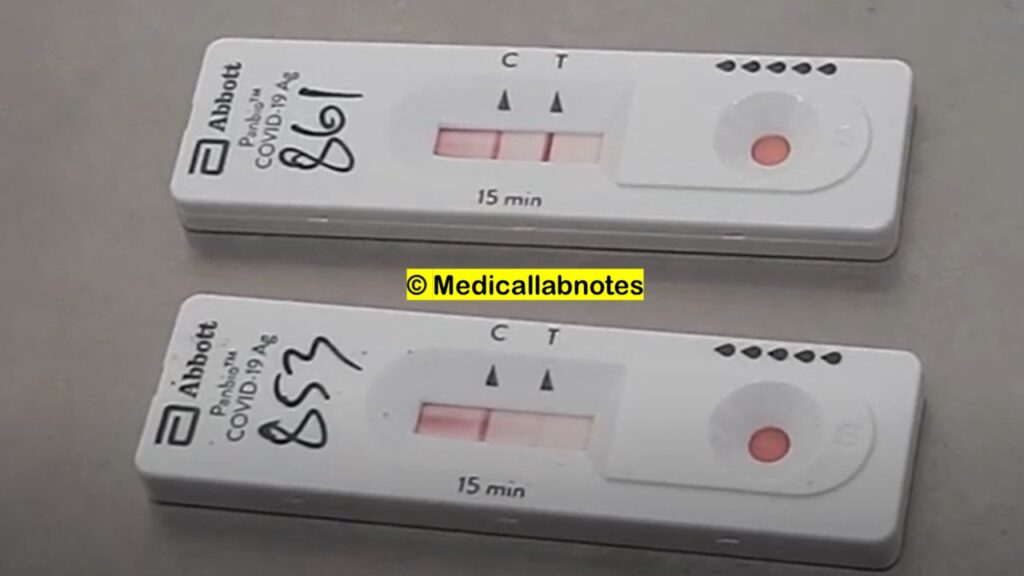
- Positive result:
- A positive result is indicated when the test device shows the presence of a visible signal, such as a colored line, in the designated area for the SARS-CoV-2 antigen.
- A positive result suggests the presence of SARS-CoV-2 viral antigens in the tested specimen, indicating an active COVID-19 infection.
- It’s important to note that a positive antigen test result should be considered preliminary and may require further confirmation with a molecular test, such as PCR, for definitive diagnosis.
- Negative result:
- A negative result is indicated when the test device shows no visible signal, or the signal is present only in the control area of the test device, and not in the designated area for the SARS-CoV-2 antigen.
- A negative result suggests the absence of detectable SARS-CoV-2 viral antigens in the tested specimen.
- However, it’s important to consider that antigen tests may have a slightly lower sensitivity compared to molecular tests like PCR. False-negative results can occur, especially during the early stages of infection or in individuals with lower viral loads. If there is a high suspicion of COVID-19 despite a negative antigen test result, a molecular test may be recommended for further confirmation.
- Invalid or inconclusive result:
- An invalid or inconclusive result may occur if the test device does not show the expected control signal, indicating that the test may have failed.
- An invalid result may be due to issues such as improper test device storage, incorrect test procedure, expired test kit, or other technical problems.
- In such cases, it’s necessary to repeat the test using a new test device and following the proper procedures.
Keynotes
Here are some keynotes on the SARS-CoV-2 antigen test:
- Rapid detection: The SARS-CoV-2 antigen test provides a rapid detection method for diagnosing active COVID-19 infections. Results can typically be obtained within minutes, allowing for quick decision-making and appropriate management.
- Point-of-care testing: Antigen tests are designed for use at the point of care, including healthcare facilities, clinics, and community testing centers. They offer convenience and accessibility, allowing for testing in various settings and reducing the need for sending samples to external laboratories.
- Screening tool: SARS-CoV-2 antigen tests can be used as a screening tool to identify individuals who may be currently infected with the virus. They can be particularly useful in situations where prompt detection and isolation of infected individuals are crucial, such as in outbreaks or high-risk settings.
- Complementary to molecular tests: Antigen tests serve as a complement to molecular tests, such as PCR, which are considered the gold standard for COVID-19 diagnosis. Antigen tests provide rapid results but have slightly lower sensitivity. Therefore, they can be used for initial screening, and positive results may require confirmation with a molecular test.
- Early infection detection: SARS-CoV-2 antigen tests can detect viral antigens during the early stages of infection when the viral load is typically high. They are particularly effective in identifying individuals who are most likely to be contagious and may transmit the virus to others.
- Limitations: It’s important to recognize the limitations of antigen tests. They may have lower sensitivity compared to molecular tests, leading to false-negative results, especially in individuals with low viral loads. Therefore, negative results should be interpreted with caution, and further testing may be required if there is a high suspicion of COVID-19.
- Public health interventions: The widespread use of SARS-CoV-2 antigen tests can help in implementing timely public health interventions, such as isolating infected individuals, contact tracing, and implementing infection control measures to prevent the spread of the virus.
- Follow manufacturer instructions: It is essential to follow the specific instructions provided by the manufacturer for the test kit being used. This includes proper sample collection techniques, correct handling of the test kit components, and accurate result interpretation.
- Regulatory approvals and guidelines: SARS-CoV-2 antigen tests should meet regulatory standards and guidelines set by health authorities in different countries. They should undergo proper validation and evaluation to ensure their accuracy and reliability.
- Ongoing research and development: The field of SARS-CoV-2 antigen testing is continuously evolving, with ongoing research and development to improve test performance, sensitivity, and specificity. New generations of antigen tests may address some of the limitations of current assays.
Further Readings
- Pilarowski, G. et al. (2020). Performance Characteristics of a Rapid SARS-CoV-2 Antigen Detection Assay at a Public Plaza Testing Site in San Francisco. Journal of Infectious Diseases, 223(7), 1139–1144. doi: 10.1093/infdis/jiaa736
- Mak, G. C. K. et al. (2020). Evaluation of Rapid Antigen Test for Detection of SARS-CoV-2 Virus. Journal of Clinical Virology, 129, 104500. doi: 10.1016/j.jcv.2020.104500
- Albert, E. et al. (2020). Field Evaluation of a Rapid Antigen Test (Panbio™ COVID-19 Ag Rapid Test Device) for COVID-19 Diagnosis in Primary Healthcare Centres. Clinical Microbiology and Infection, 27(3), 472.e7–472.e10. doi: 10.1016/j.cmi.2020.10.002
- Kevadiya, B. D. et al. (2021). Diagnosing COVID-19: The Disease and Tools for Detection. ACS Infectious Diseases, 7(3), 568–592. doi: 10.1021/acsinfecdis.0c00797
- Cerutti, F. et al. (2021). Performance of a Rapid Antigen Test in the Diagnosis of SARS-CoV-2 Infection in a Large University Hospital. Open Forum Infectious Diseases, 8(5), ofab122. doi: 10.1093/ofid/ofab122
