Introduction
Table of Contents
Stenotrophomonas maltophilia is a Gram-negative bacterium that belongs to the class Gammaproteobacteria. It is an opportunistic pathogen known for its ability to cause infections, particularly in individuals with compromised immune systems or underlying health conditions. The bacterium was initially considered to be of low clinical significance, but in recent years, it has gained attention as a cause of various infections in healthcare settings.

Key characteristics of S. maltophilia include:
- Gram-Negative Bacteria: S. maltophilia is classified as a Gram-negative bacterium based on its cell wall structure, which includes an outer membrane containing lipopolysaccharides.
- Environmental Distribution: While Stenotrophomonas maltophilia is commonly found in various environmental sources such as water, soil, plants, and hospital environments, it has been recognized as an emerging opportunistic pathogen in healthcare-associated infections.
- Antibiotic Resistance: One of the most significant challenges posed by S. maltophilia is its inherent and acquired resistance to multiple classes of antibiotics. This resistance can complicate treatment and limit available options for managing infections.
- Infections: Stenotrophomonas maltophilia infections can manifest in a variety of ways, including respiratory tract infections (pneumonia), bloodstream infections (bacteremia), urinary tract infections (UTIs), skin and soft tissue infections, and more. It is particularly problematic for individuals with weakened immune systems, such as those with cystic fibrosis, cancer patients, transplant recipients, and those on prolonged antibiotic therapy.
- Treatment Challenges: Due to its antibiotic resistance profile, treating S. maltophilia infections can be difficult. Healthcare providers often need to resort to alternative antibiotic options or combinations to achieve effective treatment. This emphasizes the importance of appropriate diagnosis and tailored treatment plans.
- Prevention and Control: Preventing Stenotrophomonas maltophilia infections in healthcare settings involves strict adherence to infection control measures, including proper hand hygiene, disinfection of equipment, and isolation protocols for infected or colonized patients.
- Research and Studies: Researchers continue to study the genetics, virulence factors, and mechanisms of antibiotic resistance in S. maltophilia to better understand how it causes infections and how it can be targeted by therapies.
Morphology
The morphology of Stenotrophomonas maltophilia refers to its physical characteristics, including its shape, size, and arrangement. Here are the key features of S. maltophilia’s morphology:

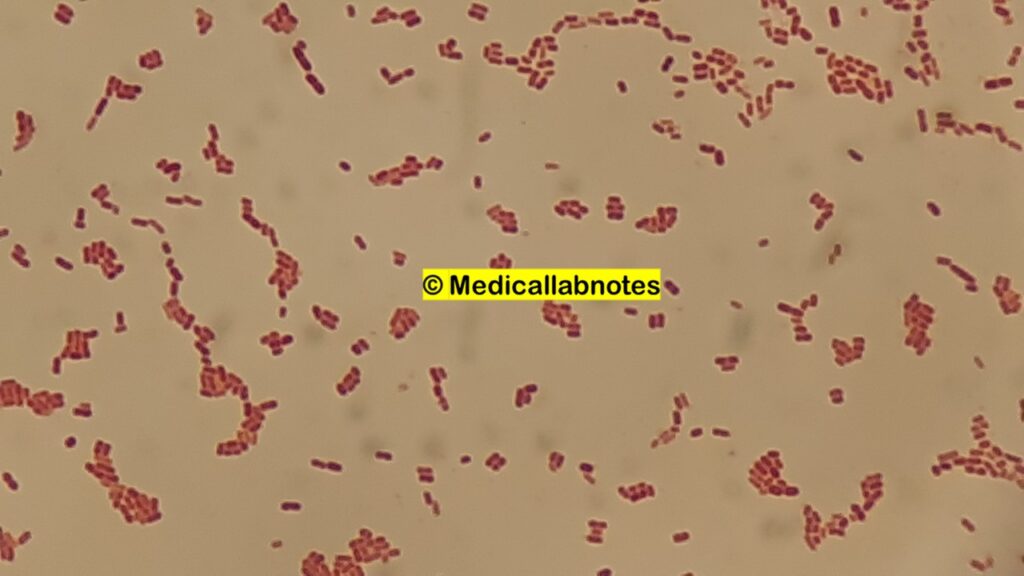
- Shape: It is a Gram-negative bacterium that typically appears as a rod-shaped or bacillus-shaped cell. These cells are cylindrical in nature and have a relatively uniform width along their length.
- Size: The size of Stenotrophomonas maltophilia cells can vary, but they generally fall within the range of 0.5 to 1.0 micrometers in width and 1.5 to 3.5 micrometers in length.
- Arrangement: Its cells can be found as single cells or arranged in pairs, short chains, or clusters. Unlike some bacterial species, S. maltophilia does not tend to form distinctive patterns like the chains seen in streptococci or the grape-like clusters seen in staphylococci.
- Motility: It is known to be motile due to the presence of polar flagella. These flagella allow the bacterium to move in liquid environments, such as water and bodily fluids.
- Color and Staining: When subjected to Gram staining, S. maltophilia will appear Gram-negative due to the structure of its cell wall. The cells will appear pink when counterstained after the initial crystal violet stain, indicating that they do not retain the crystal violet dye.
- Capsules and Biofilms: It can produce extracellular structures such as capsules and biofilms. Capsules are slimy layers that surround bacterial cells and can provide protection against immune responses and antibiotics. Biofilms are complex communities of bacteria encased in a matrix that allows them to adhere to surfaces, which can make infections more challenging to treat.
Pathogenicity
Stenotrophomonas maltophilia is considered an opportunistic pathogen, meaning that it primarily causes infections in individuals with compromised immune systems or underlying health conditions. Its pathogenicity is multifactorial, involving a combination of virulence factors and the ability to exploit vulnerabilities in the host’s defense mechanisms. Here are some key aspects of the pathogenicity of S. maltophilia:
- Virulence Factors:
- Adhesion Factors: It possesses adhesion factors that enable it to adhere to host tissues, such as epithelial cells and mucosal surfaces, which is a crucial step in establishing an infection.
- Biofilm Formation: It can form biofilms, complex communities of bacteria encased in a protective matrix. Biofilms provide protection against antibiotics and the host’s immune response, making infections harder to treat.
- Exopolysaccharides: The bacterium produces exopolysaccharides that contribute to the formation of biofilms and play a role in adherence to surfaces.
- Metallo-Beta-Lactamases (MBLs): Some strains of Stenotrophomonas maltophilia produce MBLs, which are enzymes that break down certain antibiotics, especially carbapenems, leading to antibiotic resistance.
- Antibiotic Resistance:
- S. maltophilia is notorious for its intrinsic and acquired resistance to various classes of antibiotics. This resistance includes not only beta-lactams (including carbapenems), but also aminoglycosides, fluoroquinolones, and trimethoprim-sulfamethoxazole. This makes treatment challenging and often requires the use of alternative antibiotics.
- Host Immune Suppression:
- Stenotrophomonas maltophilia infections often occur in individuals with weakened immune systems, such as those with cancer, cystic fibrosis, HIV/AIDS, organ transplants, or undergoing immunosuppressive therapy. The weakened immune response allows the bacteria to proliferate and cause infection more easily.
- Infection Sites:
- Stenotrophomonas maltophilia can cause a range of infections, including respiratory tract infections (pneumonia), bloodstream infections (bacteremia), urinary tract infections (UTIs), wound and soft tissue infections, and infections associated with medical devices like catheters and ventilators.
- Healthcare-Associated Infections:
- S. maltophilia is often associated with healthcare settings, such as hospitals and long-term care facilities. It can colonize medical equipment, surfaces, and fluids, potentially leading to infections in vulnerable patients.
- Treatment Challenges:
- Due to its antibiotic resistance and ability to form biofilms, treating Stenotrophomonas maltophilia infections can be challenging. Combination therapy and the use of less commonly used antibiotics might be necessary.
Lab Diagnosis
The laboratory diagnosis of Stenotrophomonas maltophilia infections involves a combination of microbiological techniques to isolate and identify the bacterium from clinical samples. Here are the steps typically involved in diagnosing S. maltophilia infections:

- Sample Collection:
- Clinical samples such as sputum, blood, wound swabs, urine, and respiratory secretions are collected from the patient based on the suspected site of infection.
- Sample Processing:
- The collected samples are processed to isolate the bacteria. This often involves techniques like plating the sample on agar media and incubating it under appropriate conditions.
- Culture on Selective Media:
- It can be isolated on various types of agar media, such as MacConkey agar, blood agar, and selective media specifically designed to encourage the growth of Gram-negative bacteria.
- Colonial Morphology:
- S. maltophilia colonies typically appear as non-lactose fermenting, smooth or mucoid, and slightly convex colonies on agar plates.
- Gram Staining:
- A Gram stain is performed on isolated colonies to determine the Gram-negative nature of the bacterium.
- Biochemical Tests:
- Various biochemical tests are performed to confirm the identification of Stenotrophomonas maltophilia. These tests include oxidase test, catalase test, and utilization of different sugars.
- API Identification System:
- Commercial identification systems like the API (Analytical Profile Index) system may be used to accurately identify the bacterium based on its biochemical characteristics.
- Molecular Identification:
- In some cases, molecular techniques such as polymerase chain reaction (PCR) may be employed to detect specific genetic markers unique to Stenotrophomonas maltophilia.
- Antibiotic Susceptibility Testing:
- Antibiotic susceptibility testing is crucial to determine the antibiotic resistance profile of the isolated S. maltophilia strain. This information guides the choice of appropriate antibiotics for treatment.
- MALDI-TOF Mass Spectrometry:
- Matrix-assisted laser desorption/ionization time-of-flight (MALDI-TOF) mass spectrometry is a rapid and accurate method for identifying bacterial isolates based on their protein profiles.
- Serological Tests:
- In some cases, serological tests might be used to detect specific antibodies against Stenotrophomonas maltophilia, particularly in chronic or recurrent infections.
Treatment
Treating Stenotrophomonas maltophilia infections can be challenging due to its intrinsic and acquired antibiotic resistance. The choice of treatment depends on factors such as the site and severity of infection, the patient’s overall health, and the antibiotic susceptibility profile of the specific strain. Here are some considerations and strategies for treating S. maltophilia infections:
- Antibiotic Selection:
- Trimethoprim-Sulfamethoxazole (TMP-SMX): This combination antibiotic is often considered the treatment of choice for S. maltophilia infections. However, resistance to TMP-SMX has been reported, so susceptibility testing is essential.
- Alternative Antibiotics: If TMP-SMX resistance is present, other antibiotic options may be considered. These include fluoroquinolones (e.g., levofloxacin), aminoglycosides (e.g., amikacin), and tigecycline. Combination therapy with multiple antibiotics might be necessary in some cases.
- Minocycline: In some instances, minocycline has shown activity against Stenotrophomonas maltophilia and might be considered, especially when other options are limited.
- Combination Therapy:
- Due to the bacterium’s resistance mechanisms, combination antibiotic therapy is often recommended to enhance the effectiveness of treatment. Aminoglycosides, fluoroquinolones, or tigecycline can be used in combination with other antibiotics.
- Antibiotic Sensitivity Testing:
- Accurate antibiotic susceptibility testing is crucial to guide treatment decisions. Testing should be performed to identify the most effective antibiotics against the specific strain causing the infection.
- Duration of Treatment:
- The duration of treatment varies based on the type and severity of the infection. In most cases, treatment might need to be continued for an extended period to ensure complete eradication of the bacteria.
- Dose Adjustment:
- Depending on the patient’s renal function and other factors, antibiotic dosages might need to be adjusted to achieve therapeutic levels.
- Immunomodulatory Therapy:
- Enhancing the patient’s immune response through measures such as optimizing underlying conditions, addressing immunosuppressive medications, and promoting overall health can aid in fighting the infection.
- Infection Control Measures:
- In healthcare settings, strict infection control measures are crucial to prevent the spread of Stenotrophomonas maltophilia. This includes proper hand hygiene, isolation of infected patients, and appropriate disinfection practices.
- Avoiding Unnecessary Antibiotics:
- Overuse of antibiotics can contribute to antibiotic resistance. Antibiotics should be prescribed judiciously and only when necessary.
Prevention
Preventing S. maltophilia infections, especially in healthcare settings, requires a combination of infection control measures, prudent antibiotic use, and strategies to reduce exposure to the bacterium. Here are some key prevention strategies:
- Infection Control Measures:
- Hand Hygiene: Healthcare workers should adhere to strict hand hygiene protocols, including regular handwashing with soap and water or using alcohol-based hand sanitizers.
- Isolation Precautions: Patients known to be colonized or infected with Stenotrophomonas maltophilia should be placed on appropriate isolation precautions to prevent the spread of the bacterium to other patients.
- Environmental Cleaning: Regular and thorough cleaning and disinfection of patient care areas, surfaces, and medical equipment are essential to minimize contamination.
- Personal Protective Equipment (PPE): Healthcare workers should use appropriate PPE, such as gloves and gowns, when caring for patients with S. maltophilia infections.
- Antibiotic Stewardship:
- Prudent antibiotic use is crucial to prevent the development of antibiotic-resistant strains of Stenotrophomonas maltophilia. Antibiotics should be prescribed only when necessary and based on culture and sensitivity results.
- Patient Placement:
- Patients with Stenotrophomonas maltophilia infections or colonization should be placed in single-patient rooms, if possible, to prevent cross-contamination.
- Medical Device Management:
- Proper care and maintenance of medical devices such as ventilators, urinary catheters, and central lines are important to reduce the risk of S. maltophilia infections associated with these devices.
- Water and Fluid Management:
- In healthcare settings, water sources and fluids should be regularly monitored for the presence of Stenotrophomonas maltophilia. Waterborne sources of the bacterium can be a potential reservoir for infections, especially in immunocompromised patients.
- Environmental Monitoring:
- Routine surveillance of the healthcare environment can help identify potential sources of Stenotrophomonas maltophilia and guide infection control interventions.
- Education and Training:
- Healthcare workers should receive education and training on infection prevention practices, including the importance of hand hygiene, proper use of PPE, and appropriate cleaning and disinfection techniques.
- Immunomodulation:
- For patients with underlying conditions that increase the risk of infection (such as immunosuppression or chronic lung diseases), optimizing their health and immune function can help prevent Stenotrophomonas maltophilia infections.
- Avoiding Unnecessary Procedures:
- Minimizing the use of invasive procedures and medical devices whenever possible can reduce the risk of introducing S. maltophilia into the body.
Keynotes
Here are some keynotes summarizing important aspects of Stenotrophomonas maltophilia:
- Bacterium Characteristics:
- It is a Gram-negative bacterium.
- It is rod-shaped or bacillus-shaped, typically 0.5-1.0 micrometers wide and 1.5-3.5 micrometers long.
- Motile due to polar flagella.
- Pathogenicity:
- Opportunistic pathogen causing infections in immunocompromised individuals.
- Adhesion factors, biofilm formation, and antibiotic resistance contribute to its pathogenicity.
- Infections:
- Causes a range of infections, including pneumonia, bacteremia, UTIs, and wound infections.
- Often associated with healthcare settings and medical devices.
- Antibiotic Resistance:
- Known for intrinsic and acquired resistance to multiple antibiotics, including beta-lactams, aminoglycosides, and fluoroquinolones.
- Resistance mechanisms include efflux pumps and metallo-beta-lactamases (MBLs).
- Diagnosis:
- Sample collection and culture on selective media.
- Gram staining, biochemical tests, and molecular identification techniques.
- Antibiotic susceptibility testing is crucial.
- Treatment:
- Trimethoprim-sulfamethoxazole (TMP-SMX) is often the first-line treatment.
- Combination therapy and alternative antibiotics might be necessary due to resistance.
- Treatment duration varies based on infection type and severity.
- Prevention:
- Strict infection control measures, including hand hygiene and patient isolation.
- Prudent antibiotic use and surveillance to prevent antibiotic-resistant strains.
- Proper care of medical devices and environmental monitoring.
- Research and Challenges:
- Ongoing research on virulence factors, resistance mechanisms, and treatment options.
- Challenge in managing infections due to antibiotic resistance and ability to form biofilms.
- Immunocompromised Patients:
- More susceptible to Stenotrophomonas maltophilia infections due to weakened immune systems.
- Immunomodulation and optimization of overall health can aid in preventing infections.
- Collaborative Approach:
- Prevention and management require collaboration between healthcare providers, infection control teams, and clinical microbiologists.
Further Readings
- Scientific Articles:
- Brooke, J. S. (2012). Stenotrophomonas maltophilia: an emerging global opportunistic pathogen. Clinical Microbiology Reviews, 25(1), 2-41.
- Denton, M., & Kerr, K. G. (1998). Microbiological and clinical aspects of infection associated with Stenotrophomonas maltophilia. Clinical Microbiology Reviews, 11(1), 57-80.
- Clinical Guidelines:
- Clinical and Laboratory Standards Institute (CLSI). (2021). Performance Standards for Antimicrobial Susceptibility Testing. CLSI Document M100, 31st Edition.
- Review Articles:
- Looney, W. J., & Narita, M. (2009). Maltophilia, M. (2019). Biology of the emerging pathogen Stenotrophomonas maltophilia. Clinical Microbiology Reviews, 32(3), e00038-19.
- Books:
- Mahenthiralingam, E. (Ed.). (2019). Stenotrophomonas maltophilia: Infections and Treatment. Springer.
- Online Resources:
- Centers for Disease Control and Prevention (CDC). Healthcare-Associated Infections: Stenotrophomonas maltophilia. Available at: https://www.cdc.gov/hai/organisms/stenotrophomonas.html
- Research Journals:
- Journals such as “Journal of Clinical Microbiology,” “Antimicrobial Agents and Chemotherapy,” and “Clinical Infectious Diseases” often have research articles and updates related to Stenotrophomonas maltophilia.
