Introduction
Table of Contents
The E-test, also known as the Epsilometer test, is a laboratory technique used to determine the antimicrobial susceptibility of microorganisms, particularly bacteria. It is a quantitative method that provides information about the minimum inhibitory concentration (MIC) of an antimicrobial agent against a specific organism.
The E-test utilizes strips containing a gradient of an antimicrobial agent embedded in an agar medium. These strips are placed on a plate inoculated with the test organism. As the antimicrobial agent diffuses into the surrounding agar, a concentration gradient is formed. The MIC is determined by observing the point where the bacterial growth intersects the strip, indicating the highest concentration of the antimicrobial agent that inhibits the growth of the organism.
The E-test is widely used in clinical microbiology laboratories to guide antibiotic therapy and determine the susceptibility of bacteria to various antibiotics. It provides a quantitative measurement, allowing for a more accurate interpretation and comparison of antimicrobial activity against different organisms.
The test is relatively simple to perform and provides results within 24 hours. It is a valuable tool for determining the appropriate antibiotic treatment for bacterial infections, aiding in the selection of effective drugs, and minimizing the development of antibiotic resistance.

It’s worth noting that while the E-test is primarily used for bacteria, similar techniques exist for assessing the susceptibility of other microorganisms such as fungi.
Principle
The principle of the E-test, or Epsilometer test, is based on the diffusion of an antimicrobial agent through an agar medium containing a gradient of that agent. This diffusion creates a concentration gradient that allows for the determination of the minimum inhibitory concentration (MIC) of the antimicrobial agent against a specific microorganism.
Here are the key steps and principles involved in the E-test:
- Preparation of the agar plate: A solid agar medium is prepared and spread evenly on a plate. The plate is then inoculated with a standardized suspension of the test organism, ensuring a uniform bacterial lawn.
- Application of the E-test strip: E-test strips are plastic strips with a predefined gradient of an antimicrobial agent. These strips are placed on the inoculated agar surface and gently pressed to ensure good contact.
- Diffusion of the antimicrobial agent: Once the E-test strip is applied, the antimicrobial agent starts to diffuse into the surrounding agar. As the agent diffuses, a concentration gradient is established in the agar, ranging from high concentration near the strip to low concentration further away.
- Microbial growth inhibition: The test organism, being inoculated on the agar, grows and forms a visible bacterial lawn. The growth of the organism is inhibited by the antimicrobial agent in a concentration-dependent manner.
- MIC determination: After an incubation period, typically 16 to 24 hours, the MIC is determined by observing the point where the bacterial growth intersects the E-test strip. This point represents the highest concentration of the antimicrobial agent that inhibits the growth of the organism.
- Result interpretation: The MIC value is read directly from the E-test strip. It is expressed as the concentration in micrograms per milliliter (µg/mL) or other appropriate units, depending on the specific test.

The E-test allows for the quantification of antimicrobial susceptibility, providing a numerical value for the MIC instead of a qualitative result like “susceptible” or “resistant.” This quantitative information aids in comparing different antimicrobial agents or concentrations and helps guide the selection of appropriate antibiotic therapy.
The E-test is a versatile method applicable to various microorganisms, including bacteria and fungi, and it is commonly used in clinical laboratories for antimicrobial susceptibility testing.
Purposes
The E-test, or Epsilometer test, serves several purposes in the field of microbiology and clinical practice. Here are some of its main purposes:
- Determination of Antimicrobial Susceptibility: The primary purpose of the E-test is to determine the susceptibility of microorganisms to different antimicrobial agents. By providing the minimum inhibitory concentration (MIC), the E-test helps identify the lowest concentration of an antimicrobial agent that effectively inhibits the growth of the organism. This information is crucial in guiding antibiotic therapy and selecting appropriate antibiotics for treatment.
- Antibiotic Resistance Detection: The E-test is valuable for detecting antibiotic resistance in microorganisms. By comparing the MIC obtained from the E-test with established breakpoints and interpretive criteria, it is possible to classify the organism as susceptible, intermediate, or resistant to a specific antimicrobial agent. This aids in monitoring and tracking the emergence of resistant strains, informing treatment decisions, and guiding infection control measures.
- Comparative Evaluation of Antimicrobial Agents: The E-test allows for the comparison of the effectiveness of different antimicrobial agents against a particular microorganism. By testing multiple agents simultaneously, laboratories can assess the relative potency and spectrum of activity of various antibiotics. This information assists in choosing the most appropriate and effective antibiotic for specific infections.
- Research and Epidemiological Studies: The E-test is widely used in research and epidemiological studies to investigate antimicrobial resistance patterns, track the spread of resistant strains, and analyze the effectiveness of new antimicrobial agents. It provides quantitative data that can be used to study trends, evaluate the impact of interventions, and contribute to the understanding of resistance mechanisms.
- Quality Control: The E-test is also employed as a quality control measure in clinical laboratories. It helps ensure the accuracy and reliability of antimicrobial susceptibility testing by verifying that the results fall within acceptable ranges and confirming the performance of the testing system.

Overall, the E-test plays a crucial role in guiding antibiotic therapy, detecting resistance, comparing antimicrobial agents, conducting research, and maintaining quality control in microbiology laboratories. It aids in the effective management of infectious diseases and supports efforts to combat antimicrobial resistance.
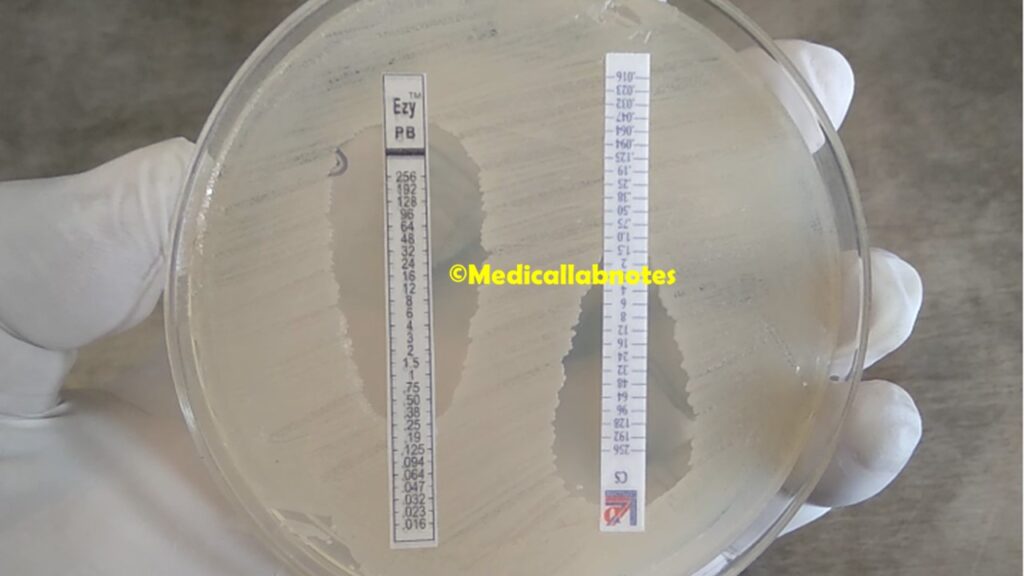
Procedure
The E-test, or Epsilometer test, follows a standard procedure to determine the antimicrobial susceptibility of microorganisms. Here is a general outline of the procedure:
- Preparation of the inoculum: Start by preparing a standardized suspension of the test organism. This involves selecting a well-isolated colony from an agar plate and suspending it in a sterile saline solution. The turbidity of the suspension should match the turbidity of a McFarland standard (usually 0.5) to ensure a consistent inoculum density.
- Inoculation of agar plate: Take a Mueller-Hinton agar plate (commonly used for bacterial susceptibility testing) and allow it to dry. Using a sterile swab, streak the agar surface with the prepared suspension of the test organism, ensuring a uniform bacterial lawn. Repeat the streaking in multiple directions for even distribution.
- Application of E-test strip: Take the E-test strip containing the antimicrobial agent of interest. The strip has a gradient of antimicrobial agents embedded in it. Carefully place the strip on the inoculated agar surface, ensuring it adheres firmly without any air bubbles or wrinkles. Gently press the strip to ensure good contact with the agar.
- Incubation: Place the inoculated and E-test strip-containing agar plate in an incubator set at the appropriate temperature and incubation time for the organism being tested. Usually, it is incubated at 35-37°C for 16-24 hours.
- Reading and interpretation: After the incubation period, remove the plate from the incubator. Observe the growth pattern of the test organism. Look for the intersection point where the bacterial growth intersects the E-test strip. This point represents the MIC, the highest concentration of the antimicrobial agent that inhibits bacterial growth. Read the MIC value from the scale on the E-test strip.
- Result reporting: Record the MIC value obtained from the E-test strip. Compare the MIC value with established interpretive criteria to classify the organism’s susceptibility as susceptible, intermediate, or resistant to the tested antimicrobial agent. Report the results in a standardized format.

It’s important to note that the specific procedures and variations may vary depending on the laboratory’s protocols, the organism being tested, and the antimicrobial agents used. Therefore, it is essential to follow the instructions provided by the manufacturer of the E-test strips and adhere to the laboratory’s guidelines and standard operating procedures.
Result-Interpretations
The interpretation of the E-test results involves comparing the obtained minimum inhibitory concentration (MIC) with established interpretive criteria. These criteria are typically provided by organizations such as the Clinical and Laboratory Standards Institute (CLSI) or the European Committee on Antimicrobial Susceptibility Testing (EUCAST). The interpretation of the E-test results helps classify the organism’s susceptibility to the tested antimicrobial agent. Here’s a general guideline for result interpretation:
- Susceptible (S): If the MIC falls below the established breakpoint for susceptibility, the organism is considered susceptible to the tested antimicrobial agent. This indicates that the concentration of the agent achieved inhibitory levels against the organism, suggesting effective treatment.
- Intermediate (I): If the MIC is within the intermediate range, it means the antimicrobial agent’s effectiveness against the organism is less predictable. Intermediate susceptibility indicates that higher doses of the agent or alternative treatment options may be necessary for optimal therapeutic outcomes. Clinical judgment and consideration of the specific infection and patient factors are important in determining appropriate treatment.
- Resistant (R): If the MIC exceeds the established breakpoint for resistance, the organism is classified as resistant to the tested antimicrobial agent. This implies that the agent’s concentration achieved with standard dosing is unlikely to inhibit the organism’s growth effectively. Alternative antimicrobial agents should be considered for treatment.
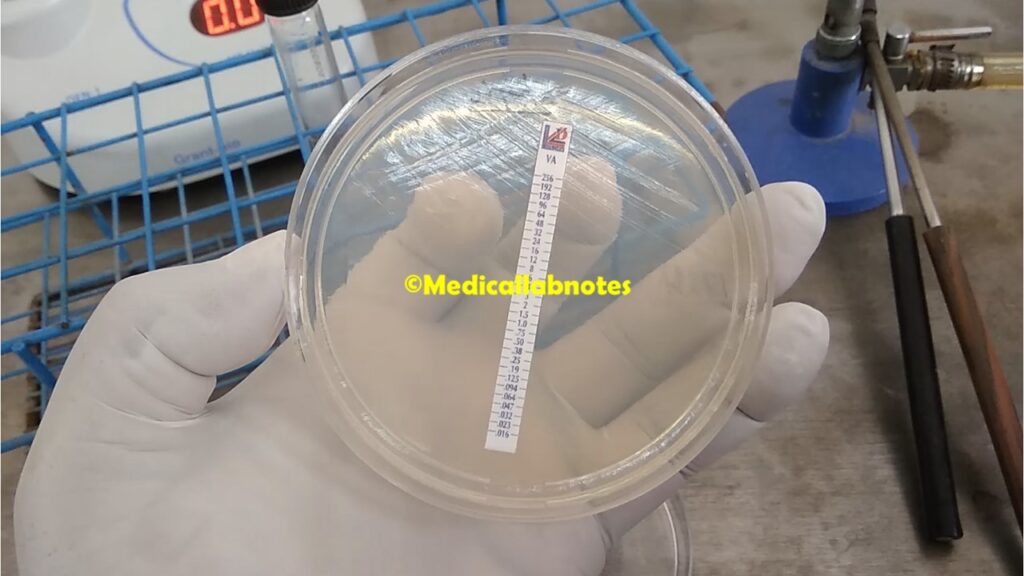
It’s important to note that the interpretive criteria, including breakpoints, may vary for different organisms and antimicrobial agents. Additionally, the interpretation should take into account the clinical context, patient factors, and any specific guidelines or recommendations from local or regional authorities.
Laboratories often provide interpretive reports that categorize the susceptibility as S, I, or R, based on the E-test results. These reports aid clinicians in making informed decisions regarding antibiotic selection and treatment strategies.
Remember to consult the specific guidelines, breakpoints, and interpretive criteria applicable to the region and the organism being tested for accurate result interpretation.
Advantages
The E-test, or Epsilometer test, offers several advantages that contribute to its widespread use in microbiology laboratories. Here are some of the key advantages of the E-test:
- Quantitative Measurement: The E-test provides a quantitative measurement of the minimum inhibitory concentration (MIC) of an antimicrobial agent. Instead of relying on qualitative results such as “susceptible” or “resistant,” the E-test offers a numerical value for the MIC. This quantitative information allows for more precise interpretation, comparison of antimicrobial activity, and better-informed treatment decisions.
- Wide Range of Antimicrobial Agents: The E-test can be performed using a broad range of antimicrobial agents, including antibiotics, antifungals, and antivirals. This versatility makes it applicable to various microorganisms and facilitates comprehensive susceptibility testing across different drug classes.
- The gradient of Concentrations: The E-test strip contains a predefined gradient of the antimicrobial agent. This gradient allows for the assessment of a wide range of concentrations simultaneously, providing a comprehensive picture of the organism’s susceptibility profile. It helps identify the exact concentration at which the agent inhibits bacterial growth, aiding in the selection of appropriate dosing regimens.
- Ease of Use: The E-test is relatively simple to perform, requiring minimal technical expertise. The test strips are easy to apply on the agar surface, and the procedure does not involve complex steps or specialized equipment. Laboratories with basic microbiological capabilities can readily adopt and perform the E-test.
- Rapid Results: The E-test offers a relatively fast turnaround time compared to some other susceptibility testing methods. Depending on the organism and incubation conditions, results can typically be obtained within 16 to 24 hours. This allows for timely decision-making in clinical settings, enabling more prompt initiation of appropriate antibiotic therapy.
- Comparative Analysis: The E-test facilitates the comparative evaluation of different antimicrobial agents. By testing multiple agents simultaneously, laboratories can assess and compare their efficacy against a particular organism. This information assists in selecting the most effective treatment options and contributes to antimicrobial stewardship efforts.
- Research and Epidemiological Applications: The quantitative nature of the E-test makes it valuable for research and epidemiological studies. It enables the analysis of resistance patterns, tracking the spread of resistant strains, and evaluating the effectiveness of new antimicrobial agents. The E-test’s consistent and reproducible results make it suitable for generating reliable data in various research settings.
- Quality Control: The E-test serves as a quality control measure for antimicrobial susceptibility testing in laboratories. It helps verify the accuracy and reliability of testing systems by providing a standardized method for testing reference strains and monitoring the performance of the laboratory’s procedures and reagents.

These advantages contribute to the widespread adoption of the E-test in clinical and research settings, making it a valuable tool for antimicrobial susceptibility testing.

Disadvantages
Here are some of the key disadvantages associated with the E-test:
- Limited Spectrum of Antimicrobial Agents: Although the E-test covers a wide range of antimicrobial agents, it may not include all available drugs. Some newer or less commonly used antimicrobial agents may not have commercially available E-test strips. This limitation restricts the comprehensive assessment of antimicrobial susceptibility for certain agents.
- Interpretation Challenges: Interpreting E-test results can sometimes be challenging due to the absence of universally accepted breakpoints for all organisms and antimicrobial agents. Interpretive criteria may vary between different organizations and regions, leading to discrepancies in result interpretation. This variability can make it difficult to compare results obtained from different laboratories or studies.
- Limited Quantitative Precision: While the E-test provides a quantitative measure of the MIC, its precision is limited compared to more advanced methods such as broth microdilution or automated systems. The MIC values obtained from the E-test may have a slightly wider range of uncertainty due to the inherent variability in strip production and visual interpretation.
- Subjectivity in Reading Results: The E-test requires visual inspection and interpretation of the growth inhibition endpoint. Human subjectivity in reading the intersection point between the strip and the bacterial growth can introduce variability in result interpretation. This subjectivity can lead to inconsistencies between different observers or laboratories.
- Dependency on Diffusion: The E-test relies on the diffusion of the antimicrobial agent through the agar medium. The rate of diffusion can vary depending on factors such as the type of agar, thickness of the agar layer, and the physicochemical properties of the agent. In some cases, slow diffusion or inadequate penetration of the agent into the agar may affect the accuracy of the MIC determination.
- Limited Application for Fastidious Organisms: The E-test may not be suitable for certain fastidious or slow-growing organisms that have specific growth requirements. The agar medium used in the E-test may not provide optimal conditions for these organisms, leading to unreliable or inconclusive results.
- Cost and Availability: The cost of E-test strips can be higher compared to other susceptibility testing methods. Additionally, the availability of specific E-test strips for rare or less common organisms may be limited, making it challenging to perform the E-test for these organisms.

It’s important to note that despite these disadvantages, the E-test remains a widely used and valuable tool for antimicrobial susceptibility testing. However, it’s essential to consider these limitations and account for them when interpreting results and making treatment decisions.
Further Readings
- Clinical and Laboratory Standards Institute (CLSI): CLSI provides guidelines and standards for laboratory testing, including antimicrobial susceptibility testing methods. Their document M100-S31: Performance Standards for Antimicrobial Susceptibility Testing provides detailed information on the E-test methodology, interpretive criteria, and quality control recommendations.
- European Committee on Antimicrobial Susceptibility Testing (EUCAST): EUCAST offers guidelines for antimicrobial susceptibility testing in Europe. Their website provides updated information on breakpoints, recommendations, and resources related to the E-test method.
- Manufacturer’s Instructions: The manufacturers of E-test strips typically provide detailed instructions for performing the test, interpreting results, and quality control procedures. These instructions offer valuable insights into the specific application of the E-test and can be found on the manufacturers’ websites or package inserts.
- Scientific Journals and Publications: Numerous scientific articles and research papers have been published on the E-test method, its applications, and comparisons with other susceptibility testing methods. Searching through databases like PubMed or Google Scholar using keywords such as “E-test,” “Epsilometer test,” or “antimicrobial susceptibility testing” will yield a wide range of articles to explore.
- Pfaller, M. A., & Koontz, F. P. (2019). Laboratory approaches for determining the susceptibility of bacteria to antimicrobial agents. In Mandell, Douglas, and Bennett’s Principles and Practice of Infectious Diseases (pp. 303-313). Elsevier.
- Van Eldere, J. (2003). Multicenter evaluation of the E test method for fluconazole susceptibility testing of Candida spp. Journal of Clinical Microbiology, 41(9), 3235-3239.
- Rennie, R. P., Turnbull, L., & Brosnikoff, C. (2007). Clostridium difficile ribotype 027 is not detected by three rapid enzyme immunoassay kits for the detection of glutamate dehydrogenase and toxin A/B. Journal of Clinical Microbiology, 45(2), 536-538.
- Kahlmeter, G., Brown, D. F., Goldstein, F. W., MacGowan, A. P., Mouton, J. W., & Österlund, A. (2003). European harmonization of MIC breakpoints for antimicrobial susceptibility testing of bacteria. Journal of Antimicrobial Chemotherapy, 52(2), 145-148.

I am no longer certain the place you are getting your info, but good topic. I must spend a while learning more or figuring out more. Thank you for excellent info I used to be in search of this info for my mission.