Introduction
Table of Contents
Streptococcus agalactiae, commonly known as Group B Streptococcus (GBS), is a Gram-positive bacterium belonging to the Streptococcaceae family. It is a significant human pathogen known for causing various infections in both adults and newborns. The bacterium is typically found as a part of the normal microbial flora in the gastrointestinal and genitourinary tracts of many healthy individuals.
Infections caused by S. agalactiae can range from mild to severe and can lead to life-threatening conditions in vulnerable populations. These include pregnant women, newborns, the elderly, and individuals with compromised immune systems. The bacterium is a leading cause of neonatal sepsis, pneumonia, and meningitis in newborns, especially during the first week of life.
Transmission of GBS usually occurs from mother to baby during childbirth, as the bacterium can be present in the genital and rectal areas of the mother. Additionally, Streptococcus agalactiae can cause infections in non-pregnant adults, such as urinary tract infections (UTIs), skin and soft tissue infections, and bloodstream infections.
Identification of GBS in clinical settings is typically done through cultures of relevant body fluids or tissues. Once diagnosed, treatment with appropriate antibiotics is crucial to control the infection and prevent complications.
In recent years, there have been significant efforts to raise awareness about Group B Streptococcus and implement preventive measures, such as universal screening of pregnant women and administering intrapartum antibiotics to reduce the risk of transmission to newborns.
Morphology
Streptococcus agalactiae, like other streptococci, exhibits a characteristic morphology that can be observed under a microscope. Here are the main morphological features of S. agalactiae:#
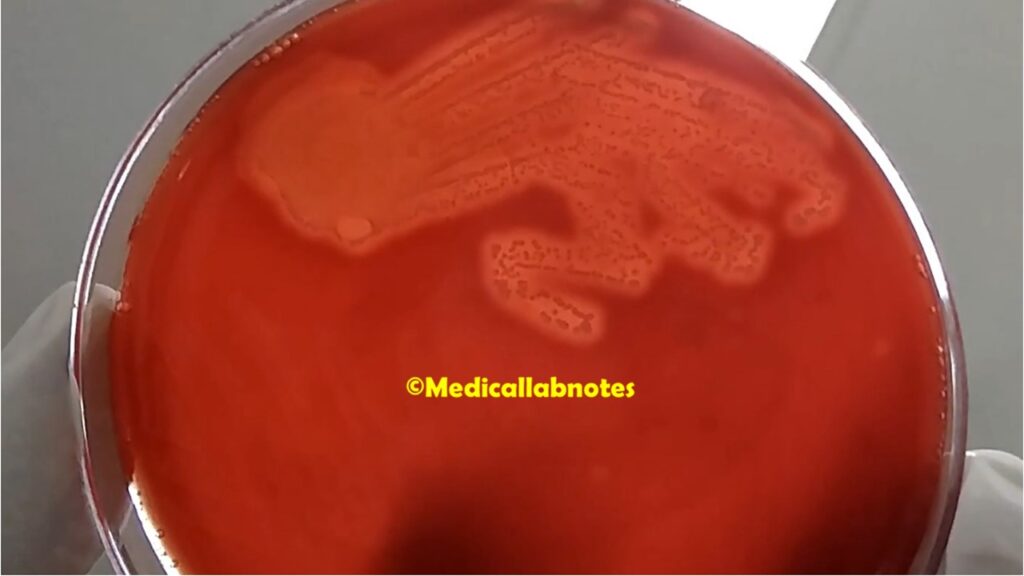
- Shape: S. agalactiae is a spherical bacterium, and its individual cells are generally round or oval-shaped.
- Arrangement: These bacteria often arrange themselves in chains, which is a distinguishing feature of streptococci. The chain formation occurs due to cell division in a single plane, resulting in a linear arrangement of cells.
- Gram Stain: It is classified as Gram-positive, meaning it retains the violet stain during Gram staining. The thick peptidoglycan layer in the cell wall of Gram-positive bacteria traps the crystal violet dye, causing the cells to appear purple under a microscope.
- Capsule: It is known for producing a polysaccharide capsule that surrounds the bacterial cells. The capsule plays a crucial role in the pathogenicity of the bacterium, as it helps in evading the host’s immune system and facilitates the attachment of bacteria to host cells.
- Size: The size of individual Streptococcus agalactiae cells typically ranges from 0.6 to 1.0 micrometers (µm) in diameter.

Pathogenicity
Streptococcus agalactiae is a pathogenic bacterium that can cause a variety of infections in both newborns and adults. Its pathogenicity is mainly attributed to several factors that allow it to colonize and invade host tissues. Some of the key aspects of the pathogenicity of S. agalactiae are as follows:
- Adherence and Colonization: Streptococcus agalactiae has mechanisms that enable it to adhere to and colonize various surfaces within the human body. For example, the bacterium can adhere to the mucous membranes of the genital and rectal areas, allowing it to establish itself in the vaginal tract of pregnant women and increasing the risk of transmission to newborns during childbirth.
- Capsule Formation: S. agalactiae produces a polysaccharide capsule, which is a key virulence factor. The capsule helps the bacterium evade the host’s immune system, making it more resistant to phagocytosis and other immune responses. This increases its ability to survive and multiply in the host’s tissues.
- Toxin Production: Some strains of Streptococcus agalactiae can produce toxins that damage host cells and contribute to the severity of infections. These toxins may lead to tissue destruction and inflammation at the site of infection.
- Hemolytic Activity: It can exhibit hemolytic activity, meaning it can lyse red blood cells. This characteristic is important in differentiating the bacterium into different serotypes, based on its hemolytic pattern.
- Invasion: Once established in the host’s tissues, S. agalactiae can invade deeper into the body, leading to various infections such as urinary tract infections, skin and soft tissue infections, and bloodstream infections.
- Neonatal Infections: It is a leading cause of neonatal sepsis, pneumonia, and meningitis. During childbirth, the bacterium can be transmitted from the mother to the newborn, leading to potentially life-threatening infections in the first week of life.
The pathogenicity of Streptococcus agalactiae can be particularly dangerous in vulnerable populations, such as pregnant women, newborns, the elderly, and individuals with weakened immune systems. Prevention and early detection are crucial to managing and controlling infections caused by this bacterium. Measures like universal screening of pregnant women and intrapartum antibiotics have been implemented to reduce the risk of transmission to newborns and mitigate the impact of S. agalactiae infections.
Lab Diagnosis
The laboratory diagnosis of Streptococcus agalactiae involves various techniques and tests to identify the bacterium accurately. Here are the common methods used in the lab diagnosis of S. agalactiae:
- Gram Staining: The initial step in identifying the bacterium is through Gram staining. Streptococcus agalactiae is Gram-positive, so it will retain the crystal violet stain and appear purple under the microscope.
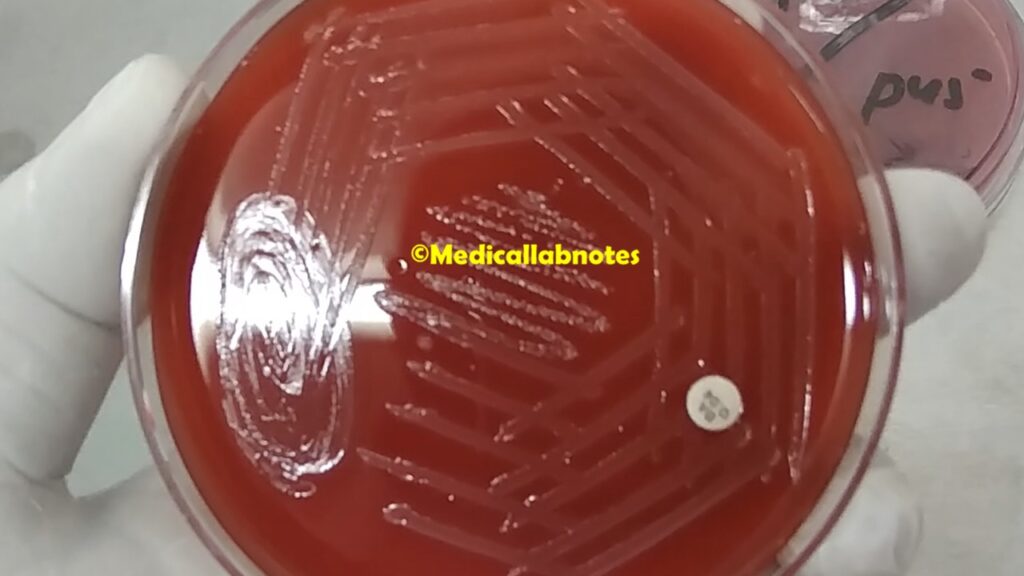
- Culture and Isolation: The most critical step is the isolation of the bacterium from the clinical sample. Common samples include vaginal/rectal swabs from pregnant women, blood cultures, cerebrospinal fluid (CSF) for neonatal meningitis, or other body fluids from infected individuals. The samples are streaked onto appropriate agar plates, such as blood agar or selective media like Columbia CNA agar, which allows the selective growth of Gram-positive bacteria.
- Hemolysis Test: It can be classified into different serotypes based on its hemolytic activity on blood agar. It produces beta-hemolysis, where it completely lyses red blood cells surrounding the colonies, resulting in a clear zone around the colonies.
- Catalase Test: Streptococcus agalactiae is catalase-negative, meaning it does not produce catalase enzyme. When hydrogen peroxide is added to a colony of Streptococcus agalactiae, it will not produce bubbles, differentiating it from catalase-positive bacteria like Staphylococcus species.
- The CAMP test is applied to the presumptive identification of Streptococcus agalactiae. It was first described in 1944 by Christie, Atkins, and Munch-Petersen, and the CAMP test is an acronym of their names. Principle of CAMP Test-The beta lysin produced by Staphylococcus aureus is sphingomyelinase in nature, which acts on the sphingomyelin present on the cell membrane of sheep red blood cells. This results in the formation of ceramide which is cleared by the action of CAMP factor i.e. extracellular diffusible protein secreted by S. agalactiae thus producing enhanced hemolysis.

- Hippurate Hydrolysis Test: S. agalactiae can hydrolyze hippurate, a compound used as a substrate in this test. Positive results will lead to the formation of a colored product, confirming the presence of the bacterium.
- Group B Antigen Test: Immunological methods, such as latex agglutination or enzyme immunoassays, can be used to detect the presence of Group B antigen specific to Streptococcus agalactiae.
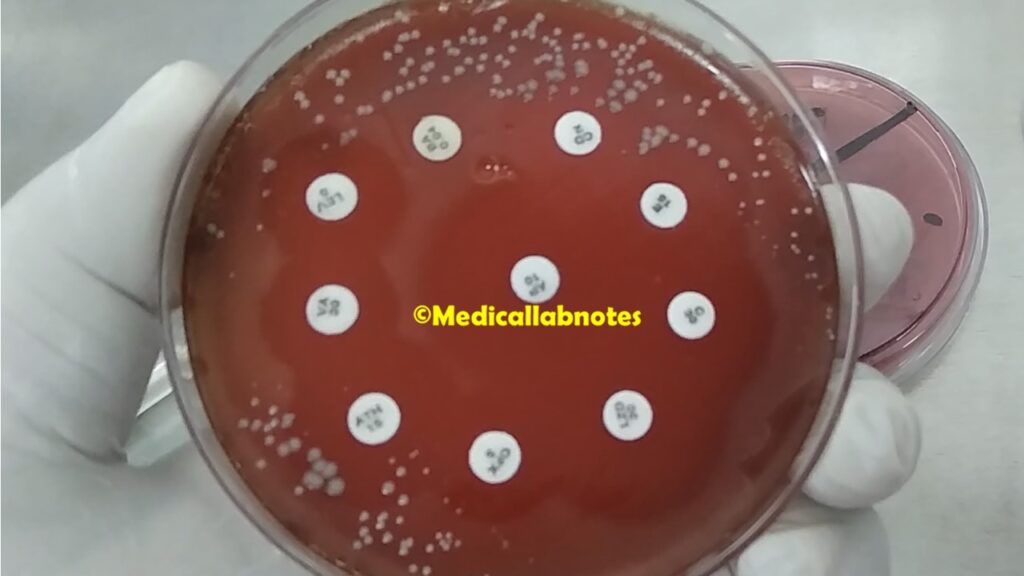
- Molecular Testing: Polymerase chain reaction (PCR) assays targeting specific genes unique to S. agalactiae can provide rapid and accurate identification of the bacterium.
Treatment
The treatment of Group B Streptococcus, GBS infections depends on the type and severity of the infection, as well as the patient’s age and overall health. Antibiotics are the mainstay of treatment for GBS infections. The choice of antibiotic may vary, and it’s essential to consider the results of antimicrobial susceptibility testing to ensure effective treatment. Here are the common treatment approaches for S. agalactiae infections:
- Intrapartum Prophylaxis: For pregnant women who are carriers of GBS, intrapartum antibiotic prophylaxis (IAP) is recommended to reduce the risk of transmission of GBS to the newborn during childbirth. Penicillin or ampicillin is the preferred antibiotic for IAP, given intravenously at appropriate intervals during labor.
- Neonatal Sepsis and Meningitis: Neonates with suspected or confirmed GBS sepsis or meningitis are treated with intravenous antibiotics. Ampicillin, often combined with gentamicin, is the first-line treatment for early-onset neonatal GBS infections. For late-onset neonatal infections, antimicrobial therapy is generally tailored based on local antimicrobial susceptibility patterns.
- Urinary Tract Infections (UTIs): For urinary tract infections caused by GBS in non-pregnant adults, oral or intravenous antibiotics are used. Common choices include penicillin, ampicillin, or ceftriaxone.
- Skin and Soft Tissue Infections: Mild skin and soft tissue infections can sometimes be managed with oral antibiotics, such as penicillin or amoxicillin. More severe cases may require intravenous antibiotics, including penicillin or ceftriaxone.
- Endocarditis and Invasive Infections: For more serious infections, such as endocarditis or infections involving deep tissues, prolonged intravenous antibiotic therapy is usually required. A combination of antibiotics may be used, based on the susceptibility of the bacterium and the clinical response of the patient.
It is essential to complete the full course of antibiotics as prescribed by the healthcare provider to ensure complete eradication of the infection and to prevent the development of antibiotic resistance. In some cases, patients may require hospitalization for intravenous antibiotics, while others can be managed with oral antibiotics as outpatients.
It’s crucial for healthcare providers to consider the patient’s medical history, allergies, and potential drug interactions when choosing the appropriate antibiotic treatment. Regular monitoring and follow-up are necessary to assess the response to treatment and ensure the infection is fully resolved.
Prevention
Preventing Streptococcus agalactiae infections is of utmost importance, especially in vulnerable populations like pregnant women and newborns. There are several preventive measures that can help reduce the transmission and impact of GBS infections:
- Universal Screening: Pregnant women should be screened for GBS colonization between 35 and 37 weeks of gestation. This screening involves taking vaginal and rectal swabs to detect the presence of GBS. Identifying colonized women allows for the implementation of intrapartum antibiotic prophylaxis (IAP) to reduce the risk of transmission to the newborn.
- Intrapartum Antibiotic Prophylaxis (IAP): For pregnant women who test positive for GBS or have an unknown colonization status but have certain risk factors, IAP is recommended. Penicillin or ampicillin is the first-line antibiotic for IAP. For those with penicillin allergy, alternative antibiotics such as clindamycin or erythromycin can be used.
- Culturing High-Risk Patients: In cases where universal screening is not available or practical, culturing high-risk patients with signs or symptoms of GBS infection may help identify colonized individuals who require IAP.
- Hospital Infection Control: Healthcare facilities should follow strict infection control practices to prevent the transmission of GBS between patients. This includes appropriate hand hygiene, cleaning and disinfection of medical equipment, and adherence to isolation precautions when necessary.
- Newborn Care: Newborns born to GBS-positive mothers should be closely monitored for signs of infection and receive appropriate medical care if symptoms arise. Proper neonatal care and early treatment can help prevent complications.
- Vaccines: Research is ongoing to develop vaccines against GBS infections. Vaccination of pregnant women to protect both the mother and newborn is a potential strategy to prevent GBS transmission and neonatal infections.
- Education and Awareness: Raising awareness among healthcare providers and the general public about the risks associated with GBS infections and the importance of preventive measures is crucial in reducing the burden of this bacterium.
Keynotes
Here are some keynotes on Streptococcus agalactiae:
- Bacterial Characteristics: S. agalactiae is a Gram-positive bacterium that appears as round or oval-shaped cells. It forms chains due to division in a single plane. It produces beta-hemolysis on blood agar, which differentiates it from other streptococci.
- Colonization and Transmission: GBS is a common inhabitant of the human gastrointestinal and genitourinary tracts. It can colonize the vagina and rectum of pregnant women, leading to vertical transmission to newborns during childbirth.
- Pathogenicity: GBS is a significant human pathogen, causing various infections such as neonatal sepsis, pneumonia, meningitis, and urinary tract infections. It produces a capsule that helps evade the host’s immune system, and some strains can produce toxins that contribute to tissue damage.
- Neonatal Infections: GBS is a leading cause of early-onset neonatal infections, usually occurring within the first week of life. Late-onset neonatal infections can also occur, often resulting from horizontal transmission.
- Intrapartum Antibiotic Prophylaxis (IAP): Universal screening of pregnant women and administration of intrapartum antibiotics to colonized women can prevent early-onset neonatal GBS infections.
- Diagnosis: GBS can be diagnosed through Gram staining, culture and isolation on selective media, and hemolysis testing. Molecular testing like PCR can provide rapid and accurate identification.
- Treatment: Antibiotics, particularly penicillin or ampicillin, are the mainstay of treatment for GBS infections. The choice of antibiotics depends on the type and severity of the infection and antimicrobial susceptibility patterns.
- Prevention: Key preventive measures include universal screening of pregnant women, IAP for GBS-positive mothers, hospital infection control practices, and educating healthcare providers and the public about GBS risks and prevention.
- Vaccines: Research is ongoing to develop vaccines against GBS infections, aiming to protect both pregnant women and newborns.
- Public Health Importance: GBS infections can be severe and life-threatening, especially in vulnerable populations. Early detection, appropriate treatment, and preventive measures are essential to reduce the impact of GBS on public health.
Further Readings
- Centers for Disease Control and Prevention (CDC) – Group B Streptococcus (GBS): Website: https://www.cdc.gov/groupbstrep/index.html The CDC provides comprehensive information about Group B Streptococcus, including epidemiology, prevention guidelines, and resources for healthcare professionals and the public.
- World Health Organization (WHO) – Streptococcus agalactiae (Group B Streptococcus) Fact Sheet: Website: https://www.who.int/news-room/fact-sheets/detail/group-b-streptococcus-(gbs) The WHO fact sheet offers a global overview of Streptococcus agalactiae, its clinical impact, and prevention strategies.
- American Academy of Pediatrics (AAP) – Guidelines for the Prevention of Perinatal Group B Streptococcal (GBS) Disease: Article: https://pediatrics.aappublications.org/content/early/2019/07/18/peds.2019-1466 This article presents the latest guidelines from the AAP for the prevention of perinatal GBS disease.
- Clinical Microbiology Reviews – Streptococcus agalactiae (Group B Streptococcus) Colonization: Epidemiology, Pathogenesis, and Clinical Significance: Article: https://cmr.asm.org/content/20/3/536 This review article provides in-depth insights into GBS colonization, pathogenesis, and its clinical significance.
- Journal of Medical Microbiology – Streptococcus agalactiae (Group B Streptococcus) Vaccines for Preventing Neonatal Infections: Article: https://jmm.microbiologyresearch.org/content/journal/jmm/10.1099/jmm.0.2008/000653-0 This paper discusses the development and potential impact of GBS vaccines in preventing neonatal infections.
- UpToDate – Group B Streptococcal Infection in Pregnant Women: Article: https://www.uptodate.com/contents/group-b-streptococcal-infection-in-pregnant-women This resource offers a detailed clinical overview of GBS infection in pregnant women, including screening and management.
- European Society of Clinical Microbiology and Infectious Diseases (ESCMID) – ESCMID Study Group for Streptococci and Streptococcal Diseases (ESSS): Website: https://www.escmid.org/research_projects/study_groups/esss/ This research group focuses on the study of streptococci and streptococcal diseases, including Streptococcus agalactiae.

I have been checking out a few of your posts and i can state pretty good stuff. I will make sure to bookmark your website.