Introduction
Table of Contents
The Xpert MTB/RIF assay is an automated, cartridge-based molecular test used to detect Mycobacterium tuberculosis (MTB) and rifampicin (RIF) resistance, the most potent first-line anti-TB drug.

Endorsed by the WHO in 2010, the Xpert MTB/RIF assay revolutionized TB diagnosis by providing results in less than two hours. It is performed on the Cepheid GeneXpert System, which integrates sample processing and real-time PCR into a single-use disposable cartridge.
Principle
The assay uses nested real-time PCR and molecular beacons to amplify and detect a specific DNA sequence of the rpoB gene.
- Target: An 81-base pair “core region” (RRDR) of the rpoB gene.
- Mechanism: Five molecular beacons (Probes A–E) are used; each is labeled with a different fluorophore. They are designed to differentiate between the wild-type (normal) sequence and mutations associated with rifampicin resistance.
Procedure
- Sample Collection: Collect at least 1 mL of raw sputum or 0.5 mL of sputum sediment.
- Liquefaction: Add twice the volume of Xpert Sample Reagent to the sample.
- Incubation: Shake vigorously and incubate at room temperature for 15 minutes (with a second shake after 10 minutes) until the sample is completely liquefied.
- Loading: Use a sterile pipette to transfer 2 mL of the treated sample into the Xpert MTB/RIF cartridge.
- Running the Test: Scan the cartridge barcode, insert the cartridge into the GeneXpert module, and start the run via the software. Results are ready in approximately 100 minutes.

Result Interpretation
| Result | Meaning |
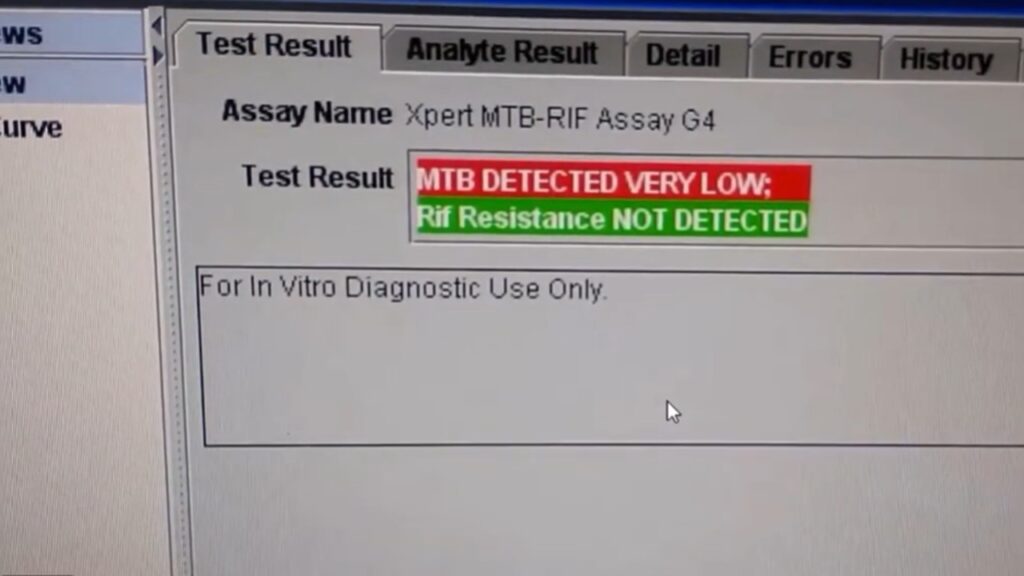
| MTB Detected; RIF Resistance Not Detected | Positive for TB; likely susceptible to rifampicin. |
| MTB Detected; RIF Resistance Detected | Positive for TB; indicates multidrug-resistant TB (MDR-TB). |
| MTB Detected; RIF Resistance Indeterminate | TB is present, but the load is too low to determine RIF status. |
| MTB Not Detected | No M. tuberculosis DNA found. |
| Invalid / Error / No Result | Test failed due to sample processing error or instrument issues; must be repeated. |
Uses
- Rapid Diagnosis: Primary diagnostic tool for individuals with clinical suspicion of pulmonary TB.
- MDR-TB Screening: Quickly identifies patients likely to have multidrug-resistant strains.
- HIV/TB Co-infection: Preferred test for people living with HIV, as they often have low bacterial loads (smear-negative).
- Infection Control: Aids in deciding if a patient should be placed in or removed from airborne isolation.
Keynotes
- Not for Monitoring: It cannot be used to monitor treatment response because it detects both live and dead DNA.
- Controls: Each cartridge includes a Sample Processing Control (SPC) to monitor PCR inhibitors and a Probe Check Control (PCC) to verify reagent integrity.
- Sensitivity: Significantly more sensitive than AFB smear microscopy, especially in smear-negative, culture-positive cases.
- Culture Still Required: Culture remains necessary for definitive drug susceptibility testing for other drugs (e.g., isoniazid).
Further Readings
- https://web-support.cepheid.com/Package%20Insert%20Files/Xpert%20MTB-RIF/Xpert%20MTB-RIF%20ENGLISH%20IFU%20303-0942%20Rev%20B.pdf
- https://redemc.net/campus/wp-content/uploads/2019/04/Implementation-of-GeneXpert-WHO-2014.pdf?x18977
- https://www.moleculartb.org/files/documents/2
- https://web-support.cepheid.com/Package%20Insert%20Files/Xpert-MTB-RIF-ENGLISH-Package-Insert-301-1404-Rev-G.pdf
- https://infomine.cepheid.com/sites/default/files/2023-07/Xpert%20MTB_RIF%20ENGLISH%20Package%20Insert%20301-0191%20Rev%20K.pdf
- https://iris.who.int/server/api/core/bitstreams/5a1e1a56-d13d-45b2-a120-f8ccc2e3c992/content
- https://ijmpr.in/article/detection-of-mycobacterium-tuberculosis-and-rifampicin-resistance-by-gene-xpert-method-among-extrapulmonary-clinical-specimens-a-cross-sectional-study-1680/
- https://www.sciencedirect.com/science/article/pii/S1876034120306055
- https://www.springermedizin.de/evaluation-of-gene-xpert-for-routine-diagnosis-of-hiv-associated/12326456
