Introduction
Table of Contents
The Xpert MTB/XDR assay is an advanced molecular diagnostic tool designed by Cepheid to rapidly detect Mycobacterium tuberculosis complex (MTBC) and its resistance to multiple first- and second-line drugs. Launched in 2021, it is categorized as a “low complexity” automated nucleic acid amplification test (NAAT).

While previous tests like Xpert MTB/RIF only identified rifampicin resistance, the Xpert MTB/XDR expands this capability to include isoniazid (INH), fluoroquinolones (FLQ), ethionamide (ETH), and second-line injectable drugs (SLIDs) like amikacin. It is primarily intended as a reflex test for patients already confirmed to have TB.
Principle
The assay uses semi-quantitative nested real-time PCR combined with High-Resolution Melting (HRM) technology.
- Detection Mechanism: It utilizes 10 “sloppy molecular beacon” (SMB) probes that target specific genomic regions.
- Melt Curve Analysis: Resistance is identified by analyzing melting temperatures (Tm). A shift in the \(Tm) compared to the wild-type (normal) sequence indicates a mutation associated with drug resistance.
- Multi-plexing: It requires a specialized 10-color GeneXpert module, unlike the 6-color modules used for standard Xpert tests.
Procedure
The test is automated after an initial manual sample preparation step and provides results in under 90 minutes.

- Sample Preparation: Mix the specimen (raw sputum or concentrated sediment) with the provided Sample Reagent (SR) at a 2:1 ratio.
- Incubation: Vigorously shake or vortex the mixture, then incubate at room temperature for 15 minutes (with occasional mixing) to ensure complete liquefaction.
- Loading: Transfer 2.0 mL of the liquefied sample into the Xpert MTB/XDR cartridge.
- Automation: Place the cartridge into the GeneXpert instrument. The system automatically performs DNA extraction, nested PCR amplification, and melt curve analysis.
Result Interpretation
The software automatically classifies results based on predefined Tm windows.
| Result | Interpretation |
| MTB Detected | M. tuberculosis complex DNA is present. |
| MTB Not Detected | No M. tuberculosis DNA found; resistance testing cannot be performed. |
| Resistance Detected | A mutation was found in the target gene for that specific drug (INH, FLQ, ETH, or SLIDs). |
| Resistance Not Detected | No mutations were found in the targeted regions for those drugs. |
| Indeterminate / Invalid | The test failed or could not definitively assess resistance for a specific drug, often due to low bacterial load. |
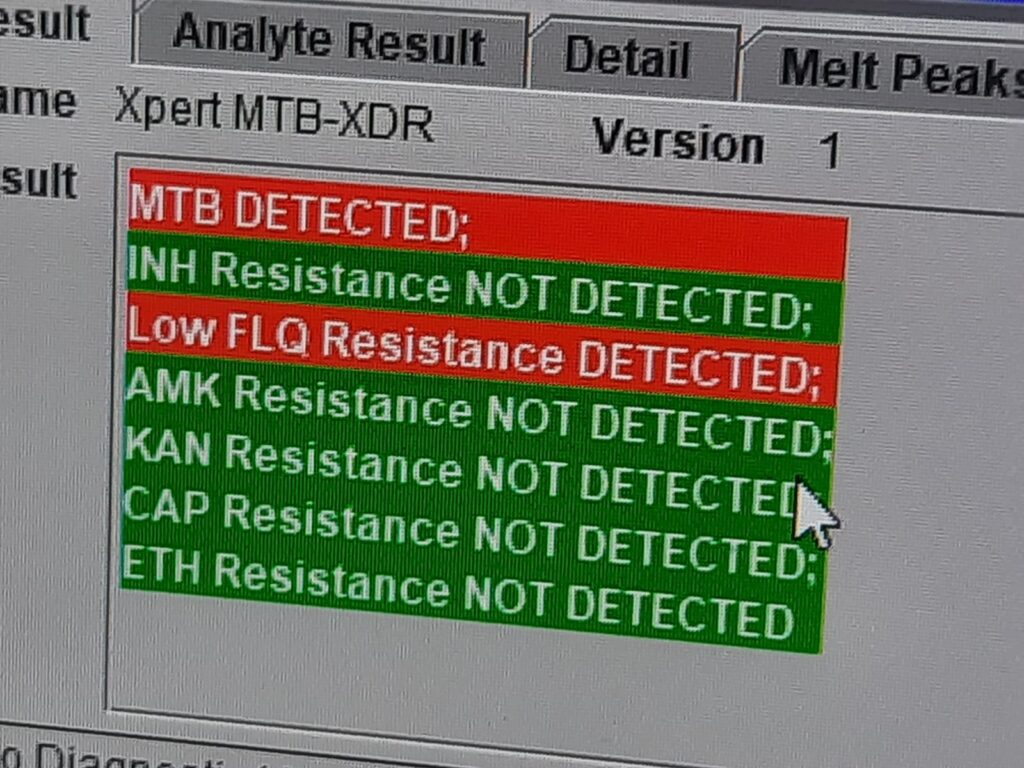
.Key Findings:
MTB DETECTED: Positive for Mycobacterium tuberculosis complex.
Low FLQ Resistance DETECTED: Indicates low-level resistance to fluoroquinolones (e.g., levofloxacin or moxifloxacin).
Resistance NOT DETECTED: No mutations were found for isoniazid (INH), amikacin (AMK), kanamycin (KAN), capreomycin (CAP), or ethionamide (ETH).
This result is consistent with a pre-XDR-TB diagnosis, as there is resistance to fluoroquinolones but susceptibility to second-line injectables.
Clinical Uses
- Treatment Planning: Allows clinicians to move away from empirical treatment by quickly providing a detailed resistance profile.
- MDR/XDR Screening: Essential for diagnosing pre-XDR-TB (MDR + FLQ resistance) and guiding all-oral treatment regimens.
- Reflex Testing: Used as a follow-on test once TB is confirmed by Xpert MTB/RIF or Ultra.
Keynotes
- Accuracy: Shows high sensitivity (>94%) for INH and FLQ resistance but lower sensitivity for Ethionamide (~25–66%) because it only targets the inhA promoter.
- High vs. Low Level Resistance: It can differentiate between low- and high-level resistance for INH and FLQs, helping determine whether high-dose INH or specific fluoroquinolones can still be used.
- Hardware Requirements: Cannot be run on standard 6-color GeneXpert machines; requires the newer 10-color modules.
- Target Genes: Targets 8 genes/regions, including katG, inhA, fabG1, and oxyR-ahpC for INH; gyrA and gyrB for FLQs; rrs and eis for SLIDs.
Further Readings
- https://publications.ersnet.org/content/erj/56/5/2003616
- https://www.cepheid.com/en-LU/tests/tb-emerging-infectious-diseases/xpert-mtb-xdr.html
- https://pmc.ncbi.nlm.nih.gov/articles/PMC9115865/
- https://www.biorxiv.org/content/10.1101/2020.09.08.288787v1.full-text
- https://www.stoptb.org/sites/default/files/imported/document/Annex_2.5_Information_Sheet_-_Xpert_MTB-XDR.pdf
- https://journals.plos.org/plosone/article/file?id=10.1371/journal.pone.0284545&type=printable
- https://www.medrxiv.org/content/10.1101/2024.10.02.24314770v1.full-text
- https://academic.oup.com/ofid/article/11/8/ofae437/7723233
- https://www.mayocliniclabs.com/test-catalog/download-setup?format=pdf&unit_code=62203
- https://tdf.org.ph/assets/img/news-and-events/bpal/Use%20of%20Xpert%20MTB%20XDR%20Assay%20for%20the%20Rapid%20Detection%20of%20DRTB%20dm2021-0501.pdf
- https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0331264
