Introduction
Table of Contents
Achromobacter xylosoxidans is a gram-negative, non-fermenting bacterium that belongs to the family Alcaligenaceae. It is an aerobic, rod-shaped bacterium known for its versatile metabolic capabilities and its widespread distribution in various environments, including soil, water, and hospital settings. Originally classified as Alcaligenes xylosoxidans, the bacterium was reclassified as A. xylosoxidans in 1999 due to genetic and phenotypic differences.
It is an opportunistic pathogen that can cause a range of infections, particularly in individuals with compromised immune systems or underlying health conditions. It is frequently associated with hospital-acquired infections, especially in patients with cystic fibrosis, chronic respiratory conditions, or those with indwelling medical devices like catheters or ventilators. It can also cause urinary tract infections, bacteremia, and skin and soft tissue infections.
This bacterium’s intrinsic resistance to various antibiotics, along with its ability to form biofilms, poses challenges in its treatment and eradication. As such, infections caused by Achromobacter xylosoxidans can be difficult to manage, necessitating a thorough understanding of its characteristics and optimal treatment strategies.
Laboratory identification of A. xylosoxidans often involves microbiological culture and biochemical tests, but more advanced techniques like molecular identification through DNA sequencing are becoming increasingly common.
Research on Achromobacter xylosoxidans is ongoing to better understand its pathogenicity, antibiotic resistance mechanisms, and potential treatment options. As with any emerging infectious agent, continued surveillance and awareness are crucial to managing and preventing its spread and associated infections.
Morphology
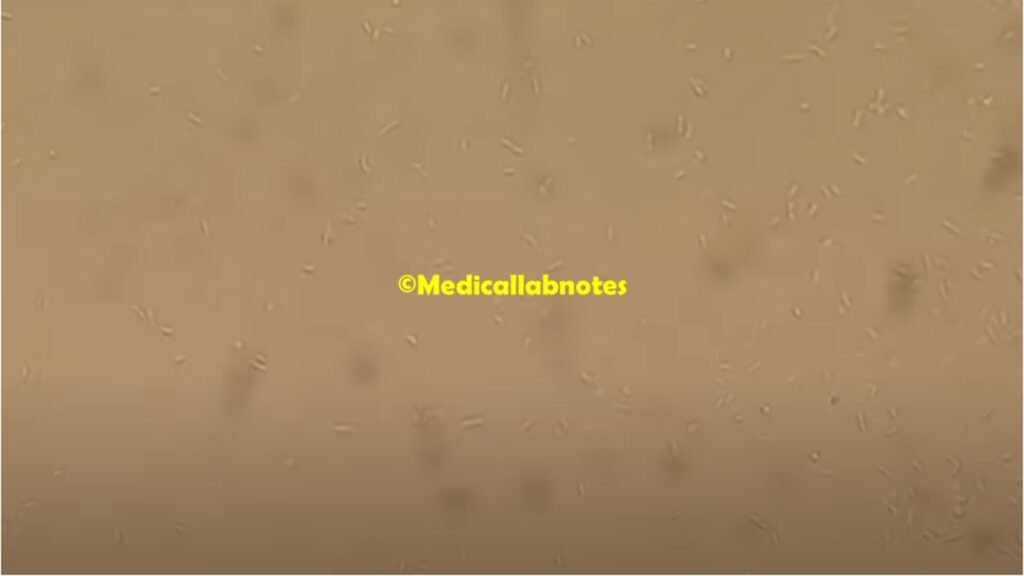
The morphology of Achromobacter xylosoxidans refers to its physical characteristics as observed under a microscope. As a gram-negative bacterium, it possesses certain distinctive features:
- Shape: It is typically rod-shaped (bacillus) and appears as straight or slightly curved rods. The length of the rods can vary.
- Size: The size of A. xylosoxidans cells can vary, but they generally fall within the range of 1.0 to 3.0 micrometers in length and 0.5 to 0.8 micrometers in width.
- Gram Staining: When subjected to the Gram staining technique, Achromobacter xylosoxidans will show a negative staining result, indicating that it does not retain the crystal violet dye and instead takes up the counterstain, safranin. This characteristic is an essential part of its identification in the laboratory.
- Motility: It is usually motile, possessing one or more flagella that allow it to move in liquid environments.
- Capsule: Some strains of Achromobacter xylosoxidans may produce a capsule, which is a slimy outer layer that aids in protecting the bacterium from the host’s immune system and other environmental factors.
Pathogenicity
Achromobacter xylosoxidans is considered an opportunistic pathogen, meaning it generally causes infections in individuals with compromised immune systems or underlying health conditions. While it can be found in various environmental sources such as soil and water, it has a particular association with hospital settings and medical devices.
Pathogenicity factors of Achromobacter xylosoxidans include:
- Biofilm Formation: A. xylosoxidans has the ability to form biofilms on medical devices like catheters, endotracheal tubes, and implanted devices. Biofilms are complex communities of bacteria encased in a protective matrix of extracellular substances, making them highly resistant to antibiotics and the host’s immune response. This allows the bacterium to persist and cause chronic infections in susceptible individuals.
- Antibiotic Resistance: Achromobacter xylosoxidans possesses inherent resistance to several classes of antibiotics. Additionally, it can acquire resistance through various genetic mechanisms, including the acquisition of resistance genes from other bacteria. This resistance complicates the treatment of infections and limits the effectiveness of commonly used antibiotics.
- Enzymatic Activity: The bacterium produces several enzymes that can contribute to tissue damage and facilitate its spread. For example, it can produce elastase, which can break down elastin, a protein in the connective tissues, and contribute to tissue destruction and inflammation.
- Endotoxin Production: Achromobacter xylosoxidans can produce endotoxins, which are toxic molecules present in the outer membrane of gram-negative bacteria. Endotoxins can trigger an inflammatory response in the host, leading to various symptoms and complications.
- Virulence Factors: While the specific virulence factors of A. xylosoxidans are not as well characterized as some other pathogens, studies have identified various potential factors that may contribute to its pathogenicity, including cell surface molecules and secreted proteins.
Achromobacter xylosoxidans infections are commonly associated with individuals with cystic fibrosis (CF) and chronic respiratory conditions, as well as patients with indwelling medical devices, such as those in intensive care units. It can cause a range of infections, including pneumonia, bloodstream infections (bacteremia), urinary tract infections (UTIs), and skin and soft tissue infections.
Lab Diagnosis
The laboratory diagnosis of Achromobacter xylosoxidans involves several steps to identify and confirm the presence of the bacterium in clinical specimens. These steps include:

- Sample Collection: The first step is to collect appropriate clinical specimens from the patient, depending on the suspected site of infection. Common specimens include sputum, blood, urine, wound swabs, or respiratory secretions.
- Microscopic Examination: The collected specimen is examined under a microscope to look for the presence of bacterial cells. A. xylosoxidans typically appears as gram-negative rods.
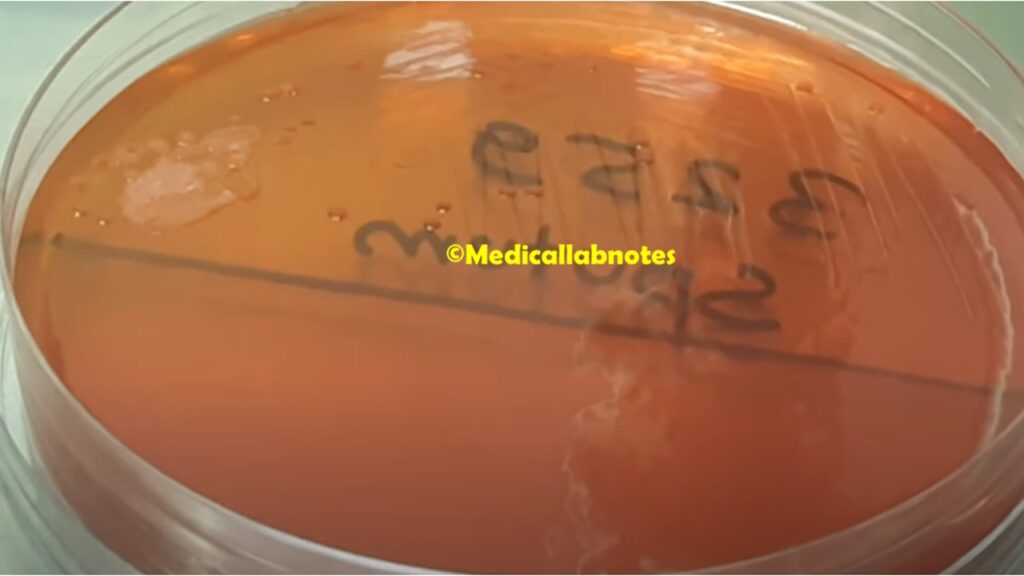
- Culture: The next step is to culture the specimen on appropriate growth media. Achromobacter xylosoxidans is aerobic, so it requires media that support the growth of aerobic bacteria. Commonly used media include MacConkey agar and blood agar.
- Identification: After the bacteria grow on the culture media, they are subjected to biochemical tests for identification. Achromobacter xylosoxidans can be distinguished from other bacteria based on its specific metabolic characteristics. Automated identification systems or manual biochemical tests may be used for this purpose.
- Gram Staining: The bacterial colonies can be subjected to Gram staining to confirm their gram-negative nature.
- MALDI-TOF Mass Spectrometry: Matrix-assisted laser desorption/ionization time-of-flight mass spectrometry (MALDI-TOF MS) is a rapid and accurate method used in many laboratories for bacterial identification, including Achromobacter xylosoxidans.
- Molecular Methods: Polymerase chain reaction (PCR) and DNA sequencing can be used for more accurate and definitive identification of Achromobacter xylosoxidans, especially in cases where traditional methods are inconclusive.
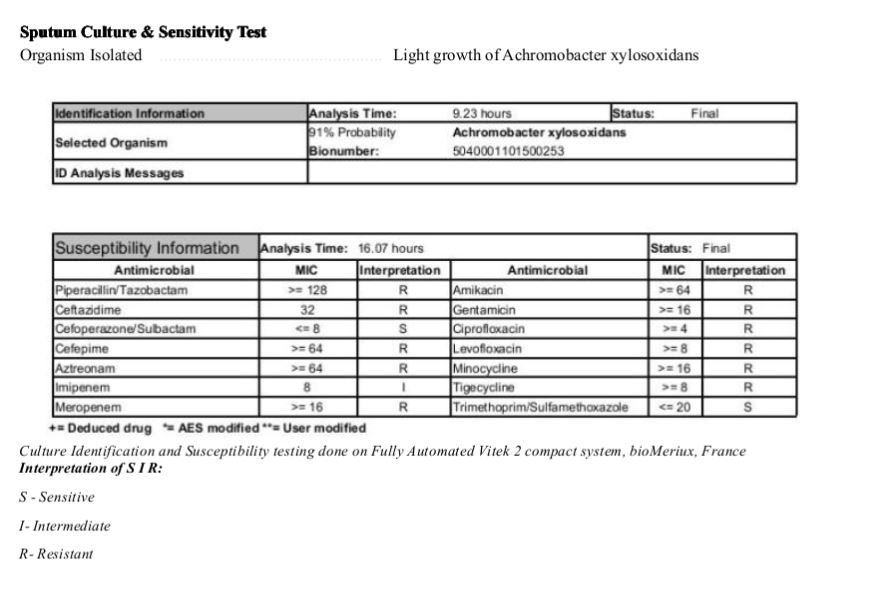
- Antimicrobial Susceptibility Testing: Once identified, the susceptibility of Achromobacter xylosoxidans to different antibiotics is determined through antimicrobial susceptibility testing. This step is crucial to guide appropriate antibiotic therapy.
Treatment
The treatment of Achromobacter xylosoxidans infections can be challenging due to its intrinsic antibiotic resistance and ability to form biofilms. The choice of treatment depends on the site and severity of the infection, as well as the individual’s overall health condition. It is essential to conduct antimicrobial susceptibility testing to determine which antibiotics are effective against the specific strain of A. xylosoxidans.
General treatment principles for Achromobacter xylosoxidans infections include:
- Combination Therapy: Achromobacter xylosoxidans is often resistant to multiple antibiotics, so combination therapy with two or more antibiotics may be necessary to achieve a synergistic effect and improve treatment efficacy. The choice of antibiotics will depend on the susceptibility testing results.
- Antibiotic Susceptibility: The selection of antibiotics should be guided by antimicrobial susceptibility testing. Some antibiotics that have been used to treat A. xylosoxidans infections include fluoroquinolones (e.g., ciprofloxacin), aminoglycosides (e.g., gentamicin), trimethoprim-sulfamethoxazole, and some beta-lactam antibiotics (e.g., piperacillin-tazobactam). However, susceptibility patterns can vary, and not all strains respond to these antibiotics.
- Duration of Treatment: The duration of antibiotic therapy depends on the type and severity of the infection. In cases of catheter-related infections or those involving medical devices, the infected device may need to be removed or replaced to effectively clear the infection.
- Biofilm Management: Achromobacter xylosoxidans‘ ability to form biofilms is a significant challenge in treatment. Biofilms are resistant to antibiotics, making it difficult for medications to penetrate and kill the bacteria. In some cases, biofilm-disrupting agents or local antimicrobial treatments may be used to improve the effectiveness of systemic antibiotics.
- Monitoring and Follow-up: Patients with A. xylosoxidans infections require close monitoring to assess treatment response and ensure appropriate adjustments in antibiotics or treatment duration if needed.
Prevention
Preventing Achromobacter xylosoxidans infections is essential, particularly in healthcare settings where vulnerable patients may be at higher risk. Here are some preventive measures to reduce the transmission and spread of A. xylosoxidans:
- Infection Control Practices: Implement and strictly adhere to infection control protocols in hospitals and healthcare facilities. This includes proper hand hygiene, regular cleaning and disinfection of patient care areas and medical equipment, and appropriate use of personal protective equipment (PPE) by healthcare workers.
- Antibiotic Stewardship: Promote responsible and judicious use of antibiotics to prevent the development of antibiotic-resistant strains of Achromobacter xylosoxidans. Avoid unnecessary or inappropriate antibiotic prescriptions and follow best practices in antibiotic administration.
- Catheter Care: Patients with indwelling medical devices like catheters are at higher risk of Achromobacter xylosoxidans infections. Proper care and maintenance of catheters are essential to minimize the risk of infection. Follow strict aseptic techniques during insertion, maintenance, and removal of catheters.
- Sterile Water: Use sterile water and appropriate disinfectants for medical procedures that involve irrigation, rinsing, or flushing to prevent contamination.
- Environmental Cleaning: Regularly clean and disinfect the environment, especially in patient rooms and shared spaces, to prevent the transmission of Achromobacter xylosoxidans between patients.
- Respiratory Hygiene: Encourage respiratory hygiene, such as covering coughs and sneezes, to minimize the spread of respiratory infections caused by A. xylosoxidans.
- Screening and Surveillance: Consider active surveillance and screening for high-risk patients, particularly those with cystic fibrosis or chronic respiratory conditions. This can help identify carriers and promptly implement infection control measures.
- Isolation Precautions: Implement appropriate isolation precautions for patients known or suspected to have Achromobacter xylosoxidans infections. This may involve placing patients in single rooms or cohorting patients with the same pathogen to prevent cross-contamination.
- Education and Training: Healthcare personnel should receive regular training on infection control practices and preventive measures, including proper hand hygiene, personal protective equipment use, and safe handling of medical devices.
- Patient Education: Educate patients and their families about Achromobacter xylosoxidans infections, transmission routes, and preventive measures they can take to reduce the risk of infection.
Keynotes
Here are some keynotes on Achromobacter xylosoxidans:
- Classification: It is a gram-negative, non-fermenting bacterium belonging to the family Alcaligenaceae.
- Morphology: It appears as rod-shaped (bacillus) bacteria under the microscope, typically measuring 1.0 to 3.0 micrometers in length and 0.5 to 0.8 micrometers in width.
- Distribution: A. xylosoxidans is widely distributed in various environments, including soil, water, and healthcare settings, particularly in patients with underlying health conditions.
- Opportunistic Pathogen: It is considered an opportunistic pathogen, causing infections in individuals with compromised immune systems or chronic medical conditions.
- Infections: Achromobacter xylosoxidans can cause a range of infections, including respiratory infections in patients with cystic fibrosis, pneumonia, urinary tract infections, bacteremia, and skin and soft tissue infections.
- Biofilm Formation: The bacterium has the ability to form biofilms on medical devices, which can make infections difficult to treat due to increased resistance to antibiotics.
- Antibiotic Resistance: Achromobacter xylosoxidans is intrinsically resistant to several antibiotics and can acquire additional resistance mechanisms, making it challenging to treat infections.
- Diagnosis: Laboratory diagnosis involves culturing the bacterium from clinical specimens, followed by identification using biochemical tests, MALDI-TOF MS, or molecular methods.
- Treatment: Treatment requires antibiotic susceptibility testing to guide appropriate antibiotic therapy. Combination therapy with multiple antibiotics is often necessary to improve effectiveness.
- Prevention: Preventive measures include infection control practices, antibiotic stewardship, proper catheter care, respiratory hygiene, environmental cleaning, screening, and isolation precautions.
- Interactions: It can interact with other bacteria and may be part of mixed infections.
- Research: Research on A. xylosoxidans is ongoing to better understand its pathogenicity, antibiotic resistance mechanisms, and optimal treatment strategies.

Further Readings
- “Achromobacter xylosoxidans in cystic fibrosis: prevalence and clinical relevance”: This study explores the prevalence and clinical impact of Achromobacter xylosoxidans in patients with cystic fibrosis. (Source: European Respiratory Journal, 2017)
- “Clinical characteristics of patients with Achromobacter xylosoxidans bacteremia: a matched case-control study”: This research investigates the clinical characteristics and outcomes of patients with Achromobacter xylosoxidans bloodstream infections. (Source: Clinical Infectious Diseases, 2010)
- “Antibiotic susceptibility patterns of Achromobacter xylosoxidans”: This study examines the antibiotic susceptibility patterns of Achromobacter xylosoxidans isolates from clinical specimens. (Source: Antimicrobial Agents and Chemotherapy, 2015)
- “Biofilm formation by Achromobacter xylosoxidans is influenced by PlcH, a novel phospholipase produced during growth on solid media”: This research explores the role of a novel phospholipase in biofilm formation by Achromobacter xylosoxidans. (Source: Infection and Immunity, 2005)
- “Mechanisms of antibiotic resistance in Achromobacter species and role of efflux pumps in resistance to carbapenems”: This study delves into the mechanisms of antibiotic resistance in Achromobacter species, with a focus on carbapenem resistance. (Source: Antimicrobial Agents and Chemotherapy, 2012)
- “Characterization of Achromobacter xylosoxidans isolates from cystic fibrosis patients: a genome-based study”: This research uses genomic approaches to characterize Achromobacter xylosoxidans isolates from individuals with cystic fibrosis. (Source: Clinical Microbiology and Infection, 2017)
- “Identification and management of Achromobacter species”: This review article provides an overview of the identification and clinical management of Achromobacter species infections. (Source: Expert Review of Anti-Infective Therapy, 2014)
